| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Farnesyl pyrophosphate synthase |
|---|
| Ligand | BDBM12578 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1914518 (CHEMBL4417101) |
|---|
| IC50 | 173±n/a nM |
|---|
| Citation |  Han, S; Li, X; Xia, Y; Yu, Z; Cai, N; Malwal, SR; Han, X; Oldfield, E; Zhang, Y Farnesyl Pyrophosphate Synthase as a Target for Drug Development: Discovery of Natural-Product-Derived Inhibitors and Their Activity in Pancreatic Cancer Cells. J Med Chem62:10867-10896 (2019) [PubMed] Article Han, S; Li, X; Xia, Y; Yu, Z; Cai, N; Malwal, SR; Han, X; Oldfield, E; Zhang, Y Farnesyl Pyrophosphate Synthase as a Target for Drug Development: Discovery of Natural-Product-Derived Inhibitors and Their Activity in Pancreatic Cancer Cells. J Med Chem62:10867-10896 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Farnesyl pyrophosphate synthase |
|---|
| Name: | Farnesyl pyrophosphate synthase |
|---|
| Synonyms: | Dimethylallyltranstransferase | FDPS | FPP synthase | FPP synthetase | FPPS_HUMAN | FPS | Farnesyl diphosphate synthase | Farnesyl diphosphate synthase (FPPS) | Farnesyl diphosphate synthetase | Farnesyl pyrophosphate synthase (FPPS) | Farnesyl pyrophosphate synthetase | Geranyltranstransferase | KIAA1293 | P14324 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 48272.89 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P14324 |
|---|
| Residue: | 419 |
|---|
| Sequence: | MPLSRWLRSVGVFLLPAPYWAPRERWLGSLRRPSLVHGYPVLAWHSARCWCQAWTEEPRA
LCSSLRMNGDQNSDVYAQEKQDFVQHFSQIVRVLTEDEMGHPEIGDAIARLKEVLEYNAI
GGKYNRGLTVVVAFRELVEPRKQDADSLQRAWTVGWCVELLQAFFLVADDIMDSSLTRRG
QICWYQKPGVGLDAINDANLLEACIYRLLKLYCREQPYYLNLIELFLQSSYQTEIGQTLD
LLTAPQGNVDLVRFTEKRYKSIVKYKTAFYSFYLPIAAAMYMAGIDGEKEHANAKKILLE
MGEFFQIQDDYLDLFGDPSVTGKIGTDIQDNKCSWLVVQCLQRATPEQYQILKENYGQKE
AEKVARVKALYEELDLPAVFLQYEEDSYSHIMALIEQYAAPLPPAVFLGLARKIYKRRK
|
|
|
|---|
| BDBM12578 |
|---|
| n/a |
|---|
| Name | BDBM12578 |
|---|
| Synonyms: | 2-(imidazol-1-yl)-1-hydroxyethylidene-1,1-bisphosphonic acid | Bisphosphonate 3 | CGP-42446 | CHEMBL924 | JMC515594 Compound 55 | Reclast | US11279719, Example Zolendronic acid (ZOL) | ZOL | Zoledronate | Zometa | [1-hydroxy-2-(1H-imidazol-1-yl)-1-phosphonoethyl]phosphonic acid | zoledronic acid |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C5H10N2O7P2 |
|---|
| Mol. Mass. | 272.0896 |
|---|
| SMILES | OC(Cn1ccnc1)(P(O)(O)=O)P(O)(O)=O |
|---|
| Structure | 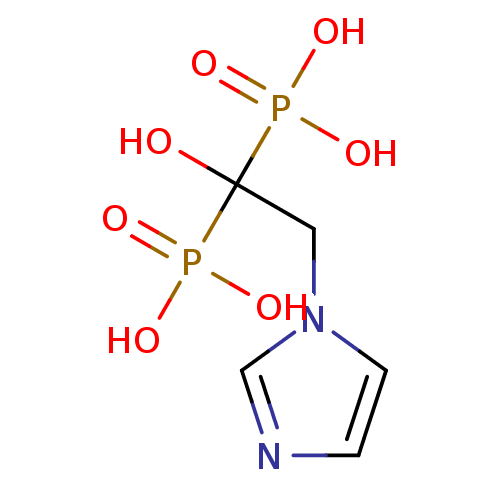
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Han, S; Li, X; Xia, Y; Yu, Z; Cai, N; Malwal, SR; Han, X; Oldfield, E; Zhang, Y Farnesyl Pyrophosphate Synthase as a Target for Drug Development: Discovery of Natural-Product-Derived Inhibitors and Their Activity in Pancreatic Cancer Cells. J Med Chem62:10867-10896 (2019) [PubMed] Article
Han, S; Li, X; Xia, Y; Yu, Z; Cai, N; Malwal, SR; Han, X; Oldfield, E; Zhang, Y Farnesyl Pyrophosphate Synthase as a Target for Drug Development: Discovery of Natural-Product-Derived Inhibitors and Their Activity in Pancreatic Cancer Cells. J Med Chem62:10867-10896 (2019) [PubMed] Article