| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50540670 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1982571 (CHEMBL4615833) |
|---|
| IC50 | >30000±n/a nM |
|---|
| Citation |  Malancona, S; Mori, M; Fezzardi, P; Santoriello, M; Basta, A; Nibbio, M; Kovalenko, L; Speziale, R; Battista, MR; Cellucci, A; Gennari, N; Monteagudo, E; Di Marco, A; Giannini, A; Sharma, R; Pires, M; Real, E; Zazzi, M; Dasso Lang, MC; De Forni, D; Saladini, F; Mely, Y; Summa, V; Harper, S; Botta, M 5,6-Dihydroxypyrimidine Scaffold to Target HIV-1 Nucleocapsid Protein. ACS Med Chem Lett11:766-772 (2020) [PubMed] Article Malancona, S; Mori, M; Fezzardi, P; Santoriello, M; Basta, A; Nibbio, M; Kovalenko, L; Speziale, R; Battista, MR; Cellucci, A; Gennari, N; Monteagudo, E; Di Marco, A; Giannini, A; Sharma, R; Pires, M; Real, E; Zazzi, M; Dasso Lang, MC; De Forni, D; Saladini, F; Mely, Y; Summa, V; Harper, S; Botta, M 5,6-Dihydroxypyrimidine Scaffold to Target HIV-1 Nucleocapsid Protein. ACS Med Chem Lett11:766-772 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50540670 |
|---|
| n/a |
|---|
| Name | BDBM50540670 |
|---|
| Synonyms: | CHEMBL4633550 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H21ClN4O3 |
|---|
| Mol. Mass. | 376.837 |
|---|
| SMILES | C[C@@H](NC(=O)c1nc(nc(O)c1O)-c1cc(Cl)ccn1)C1CCCCC1 |r| |
|---|
| Structure | 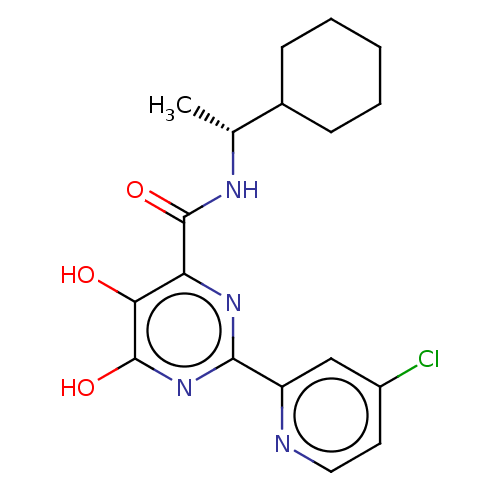
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Malancona, S; Mori, M; Fezzardi, P; Santoriello, M; Basta, A; Nibbio, M; Kovalenko, L; Speziale, R; Battista, MR; Cellucci, A; Gennari, N; Monteagudo, E; Di Marco, A; Giannini, A; Sharma, R; Pires, M; Real, E; Zazzi, M; Dasso Lang, MC; De Forni, D; Saladini, F; Mely, Y; Summa, V; Harper, S; Botta, M 5,6-Dihydroxypyrimidine Scaffold to Target HIV-1 Nucleocapsid Protein. ACS Med Chem Lett11:766-772 (2020) [PubMed] Article
Malancona, S; Mori, M; Fezzardi, P; Santoriello, M; Basta, A; Nibbio, M; Kovalenko, L; Speziale, R; Battista, MR; Cellucci, A; Gennari, N; Monteagudo, E; Di Marco, A; Giannini, A; Sharma, R; Pires, M; Real, E; Zazzi, M; Dasso Lang, MC; De Forni, D; Saladini, F; Mely, Y; Summa, V; Harper, S; Botta, M 5,6-Dihydroxypyrimidine Scaffold to Target HIV-1 Nucleocapsid Protein. ACS Med Chem Lett11:766-772 (2020) [PubMed] Article