| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase EZH2 |
|---|
| Ligand | BDBM50541900 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1985527 (CHEMBL4618933) |
|---|
| IC50 | 19±n/a nM |
|---|
| Citation |  Khanna, A; C�t�, A; Arora, S; Moine, L; Gehling, VS; Brenneman, J; Cantone, N; Stuckey, JI; Apte, S; Ramakrishnan, A; Bruderek, K; Bradley, WD; Audia, JE; Cummings, RT; Sims, RJ; Trojer, P; Levell, JR Design, Synthesis, and Pharmacological Evaluation of Second Generation EZH2 Inhibitors with Long Residence Time. ACS Med Chem Lett11:1205-1212 (2020) [PubMed] Article Khanna, A; C�t�, A; Arora, S; Moine, L; Gehling, VS; Brenneman, J; Cantone, N; Stuckey, JI; Apte, S; Ramakrishnan, A; Bruderek, K; Bradley, WD; Audia, JE; Cummings, RT; Sims, RJ; Trojer, P; Levell, JR Design, Synthesis, and Pharmacological Evaluation of Second Generation EZH2 Inhibitors with Long Residence Time. ACS Med Chem Lett11:1205-1212 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase EZH2 |
|---|
| Name: | Histone-lysine N-methyltransferase EZH2 |
|---|
| Synonyms: | ENX-1 | EZH2 | EZH2_HUMAN | Enhancer of zeste homolog 2 (EZH2) | Histone-lysine N-methyltransferase EZH2 | KMT6 | Lysine N-methyltransferase 6 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 85367.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15910 |
|---|
| Residue: | 746 |
|---|
| Sequence: | MGQTGKKSEKGPVCWRKRVKSEYMRLRQLKRFRRADEVKSMFSSNRQKILERTEILNQEW
KQRRIQPVHILTSVSSLRGTRECSVTSDLDFPTQVIPLKTLNAVASVPIMYSWSPLQQNF
MVEDETVLHNIPYMGDEVLDQDGTFIEELIKNYDGKVHGDRECGFINDEIFVELVNALGQ
YNDDDDDDDGDDPEEREEKQKDLEDHRDDKESRPPRKFPSDKIFEAISSMFPDKGTAEEL
KEKYKELTEQQLPGALPPECTPNIDGPNAKSVQREQSLHSFHTLFCRRCFKYDCFLHPFH
ATPNTYKRKNTETALDNKPCGPQCYQHLEGAKEFAAALTAERIKTPPKRPGGRRRGRLPN
NSSRPSTPTINVLESKDTDSDREAGTETGGENNDKEEEEKKDETSSSSEANSRCQTPIKM
KPNIEPPENVEWSGAEASMFRVLIGTYYDNFCAIARLIGTKTCRQVYEFRVKESSIIAPA
PAEDVDTPPRKKKRKHRLWAAHCRKIQLKKDGSSNHVYNYQPCDHPRQPCDSSCPCVIAQ
NFCEKFCQCSSECQNRFPGCRCKAQCNTKQCPCYLAVRECDPDLCLTCGAADHWDSKNVS
CKNCSIQRGSKKHLLLAPSDVAGWGIFIKDPVQKNEFISEYCGEIISQDEADRRGKVYDK
YMCSFLFNLNNDFVVDATRKGNKIRFANHSVNPNCYAKVMMVNGDHRIGIFAKRAIQTGE
ELFFDYRYSQADALKYVGIEREMEIP
|
|
|
|---|
| BDBM50541900 |
|---|
| n/a |
|---|
| Name | BDBM50541900 |
|---|
| Synonyms: | CHEMBL4633598 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H33F3N4O3S |
|---|
| Mol. Mass. | 550.636 |
|---|
| SMILES | C[C@H](C1CCN(CC(F)(F)F)CC1)n1c(C)c(C(=O)NCc2c(cc(C)[nH]c2=O)[S+](C)[O-])c2ccccc12 |r| |
|---|
| Structure | 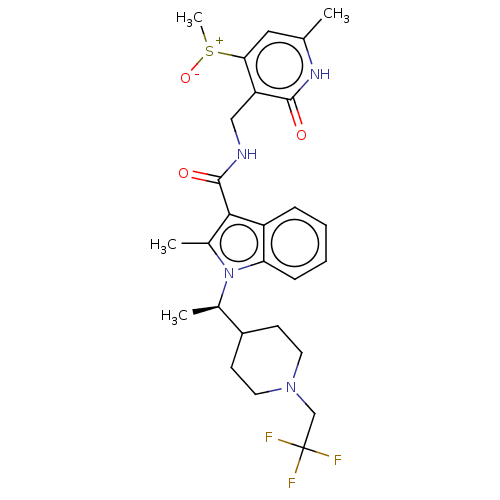
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Khanna, A; C�t�, A; Arora, S; Moine, L; Gehling, VS; Brenneman, J; Cantone, N; Stuckey, JI; Apte, S; Ramakrishnan, A; Bruderek, K; Bradley, WD; Audia, JE; Cummings, RT; Sims, RJ; Trojer, P; Levell, JR Design, Synthesis, and Pharmacological Evaluation of Second Generation EZH2 Inhibitors with Long Residence Time. ACS Med Chem Lett11:1205-1212 (2020) [PubMed] Article
Khanna, A; C�t�, A; Arora, S; Moine, L; Gehling, VS; Brenneman, J; Cantone, N; Stuckey, JI; Apte, S; Ramakrishnan, A; Bruderek, K; Bradley, WD; Audia, JE; Cummings, RT; Sims, RJ; Trojer, P; Levell, JR Design, Synthesis, and Pharmacological Evaluation of Second Generation EZH2 Inhibitors with Long Residence Time. ACS Med Chem Lett11:1205-1212 (2020) [PubMed] Article