| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor alpha |
|---|
| Ligand | BDBM50100441 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_220899 (CHEMBL824654) |
|---|
| IC50 | 10596±n/a nM |
|---|
| Citation |  Brooks, DA; Etgen, GJ; Rito, CJ; Shuker, AJ; Dominianni, SJ; Warshawsky, AM; Ardecky, R; Paterniti, JR; Tyhonas, J; Karanewsky, DS; Kauffman, RF; Broderick, CL; Oldham, BA; Montrose-Rafizadeh, C; Winneroski, LL; Faul, MM; McCarthy, JR Design and synthesis of 2-methyl-2-[4-(2-[5-methyl-2-aryloxazol-4-yl]ethoxy)phenoxy]propionic acids: a new class of dual PPARalpha/gamma agonists. J Med Chem44:2061-4 (2001) [PubMed] Brooks, DA; Etgen, GJ; Rito, CJ; Shuker, AJ; Dominianni, SJ; Warshawsky, AM; Ardecky, R; Paterniti, JR; Tyhonas, J; Karanewsky, DS; Kauffman, RF; Broderick, CL; Oldham, BA; Montrose-Rafizadeh, C; Winneroski, LL; Faul, MM; McCarthy, JR Design and synthesis of 2-methyl-2-[4-(2-[5-methyl-2-aryloxazol-4-yl]ethoxy)phenoxy]propionic acids: a new class of dual PPARalpha/gamma agonists. J Med Chem44:2061-4 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor alpha |
|---|
| Name: | Peroxisome proliferator-activated receptor alpha |
|---|
| Synonyms: | NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52222.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07869 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSC
PGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACE
GCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSE
KAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFV
IHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANL
DLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFD
FAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDI
FLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
|
|
|
|---|
| BDBM50100441 |
|---|
| n/a |
|---|
| Name | BDBM50100441 |
|---|
| Synonyms: | CHEMBL294807 | {4-[2-(5-Methyl-2-phenyl-oxazol-4-yl)-ethoxy]-phenoxy}-acetic acid |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H19NO5 |
|---|
| Mol. Mass. | 353.3686 |
|---|
| SMILES | Cc1oc(nc1CCOc1ccc(OCC(O)=O)cc1)-c1ccccc1 |
|---|
| Structure | 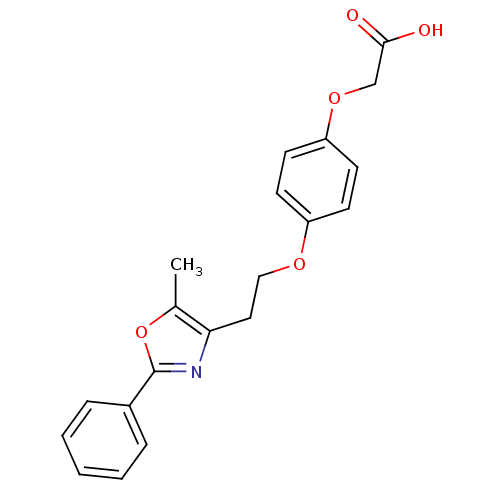
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Brooks, DA; Etgen, GJ; Rito, CJ; Shuker, AJ; Dominianni, SJ; Warshawsky, AM; Ardecky, R; Paterniti, JR; Tyhonas, J; Karanewsky, DS; Kauffman, RF; Broderick, CL; Oldham, BA; Montrose-Rafizadeh, C; Winneroski, LL; Faul, MM; McCarthy, JR Design and synthesis of 2-methyl-2-[4-(2-[5-methyl-2-aryloxazol-4-yl]ethoxy)phenoxy]propionic acids: a new class of dual PPARalpha/gamma agonists. J Med Chem44:2061-4 (2001) [PubMed]
Brooks, DA; Etgen, GJ; Rito, CJ; Shuker, AJ; Dominianni, SJ; Warshawsky, AM; Ardecky, R; Paterniti, JR; Tyhonas, J; Karanewsky, DS; Kauffman, RF; Broderick, CL; Oldham, BA; Montrose-Rafizadeh, C; Winneroski, LL; Faul, MM; McCarthy, JR Design and synthesis of 2-methyl-2-[4-(2-[5-methyl-2-aryloxazol-4-yl]ethoxy)phenoxy]propionic acids: a new class of dual PPARalpha/gamma agonists. J Med Chem44:2061-4 (2001) [PubMed]