| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Receptor tyrosine-protein kinase erbB-2 |
|---|
| Ligand | BDBM50114250 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_88298 |
|---|
| IC50 | 4.2±n/a nM |
|---|
| Citation |  Dumas, J; Hatoum-Mokdad, H; Sibley, RN; Smith, RA; Scott, WJ; Khire, U; Lee, W; Wood, J; Wolanin, D; Cooley, J; Bankston, D; Redman, AM; Schoenleber, R; Caringal, Y; Gunn, D; Romero, R; Osterhout, M; Paulsen, H; Housley, TJ; Wilhelm, SM; Pirro, J; Chien, DS; Ranges, GE; Shrikhande, A; Muzsi, A; Bortolon, E; Wakefield, J; Gianpaolo Ostravage, C; Bhargava, A; Chau, T Synthesis and pharmacological characterization of a potent, orally active p38 kinase inhibitor. Bioorg Med Chem Lett12:1559-62 (2002) [PubMed] Dumas, J; Hatoum-Mokdad, H; Sibley, RN; Smith, RA; Scott, WJ; Khire, U; Lee, W; Wood, J; Wolanin, D; Cooley, J; Bankston, D; Redman, AM; Schoenleber, R; Caringal, Y; Gunn, D; Romero, R; Osterhout, M; Paulsen, H; Housley, TJ; Wilhelm, SM; Pirro, J; Chien, DS; Ranges, GE; Shrikhande, A; Muzsi, A; Bortolon, E; Wakefield, J; Gianpaolo Ostravage, C; Bhargava, A; Chau, T Synthesis and pharmacological characterization of a potent, orally active p38 kinase inhibitor. Bioorg Med Chem Lett12:1559-62 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Receptor tyrosine-protein kinase erbB-2 |
|---|
| Name: | Receptor tyrosine-protein kinase erbB-2 |
|---|
| Synonyms: | 2.7.10.1 | C-erbB-2 | CD_antigen=CD340 | ERBB2 | ERBB2_HUMAN | ErbB-2/ErbB-3 heterodimer | FASN/HER2 | HER-2 Substrate | HER2 | MLN 19 | MLN19 | Metastatic lymph node gene 19 protein | NEU | NGL | Proto-oncogene Neu | Proto-oncogene c-ErbB-2 | Tyrosine kinase-type cell surface receptor HER2 | p185erbB2 |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 137894.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04626 |
|---|
| Residue: | 1255 |
|---|
| Sequence: | MELAALCRWGLLLALLPPGAASTQVCTGTDMKLRLPASPETHLDMLRHLYQGCQVVQGNL
ELTYLPTNASLSFLQDIQEVQGYVLIAHNQVRQVPLQRLRIVRGTQLFEDNYALAVLDNG
DPLNNTTPVTGASPGGLRELQLRSLTEILKGGVLIQRNPQLCYQDTILWKDIFHKNNQLA
LTLIDTNRSRACHPCSPMCKGSRCWGESSEDCQSLTRTVCAGGCARCKGPLPTDCCHEQC
AAGCTGPKHSDCLACLHFNHSGICELHCPALVTYNTDTFESMPNPEGRYTFGASCVTACP
YNYLSTDVGSCTLVCPLHNQEVTAEDGTQRCEKCSKPCARVCYGLGMEHLREVRAVTSAN
IQEFAGCKKIFGSLAFLPESFDGDPASNTAPLQPEQLQVFETLEEITGYLYISAWPDSLP
DLSVFQNLQVIRGRILHNGAYSLTLQGLGISWLGLRSLRELGSGLALIHHNTHLCFVHTV
PWDQLFRNPHQALLHTANRPEDECVGEGLACHQLCARGHCWGPGPTQCVNCSQFLRGQEC
VEECRVLQGLPREYVNARHCLPCHPECQPQNGSVTCFGPEADQCVACAHYKDPPFCVARC
PSGVKPDLSYMPIWKFPDEEGACQPCPINCTHSCVDLDDKGCPAEQRASPLTSIISAVVG
ILLVVVLGVVFGILIKRRQQKIRKYTMRRLLQETELVEPLTPSGAMPNQAQMRILKETEL
RKVKVLGSGAFGTVYKGIWIPDGENVKIPVAIKVLRENTSPKANKEILDEAYVMAGVGSP
YVSRLLGICLTSTVQLVTQLMPYGCLLDHVRENRGRLGSQDLLNWCMQIAKGMSYLEDVR
LVHRDLAARNVLVKSPNHVKITDFGLARLLDIDETEYHADGGKVPIKWMALESILRRRFT
HQSDVWSYGVTVWELMTFGAKPYDGIPAREIPDLLEKGERLPQPPICTIDVYMIMVKCWM
IDSECRPRFRELVSEFSRMARDPQRFVVIQNEDLGPASPLDSTFYRSLLEDDDMGDLVDA
EEYLVPQQGFFCPDPAPGAGGMVHHRHRSSSTRSGGGDLTLGLEPSEEEAPRSPLAPSEG
AGSDVFDGDLGMGAAKGLQSLPTHDPSPLQRYSEDPTVPLPSETDGYVAPLTCSPQPEYV
NQPDVRPQPPSPREGPLPAARPAGATLERPKTLSPGKNGVVKDVFAFGGAVENPEYLTPQ
GGAAPQPHPPPAFSPAFDNLYYWDQDPPERGAPPSTFKGTPTAENPEYLGLDVPV
|
|
|
|---|
| BDBM50114250 |
|---|
| n/a |
|---|
| Name | BDBM50114250 |
|---|
| Synonyms: | 1-(5-tert-Butyl-2-methyl-2H-pyrazol-3-yl)-3-(4-pyridin-4-ylmethyl-phenyl)-urea | CHEMBL288812 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H25N5O |
|---|
| Mol. Mass. | 363.4561 |
|---|
| SMILES | Cn1nc(cc1NC(=O)Nc1ccc(Cc2ccncc2)cc1)C(C)(C)C |
|---|
| Structure | 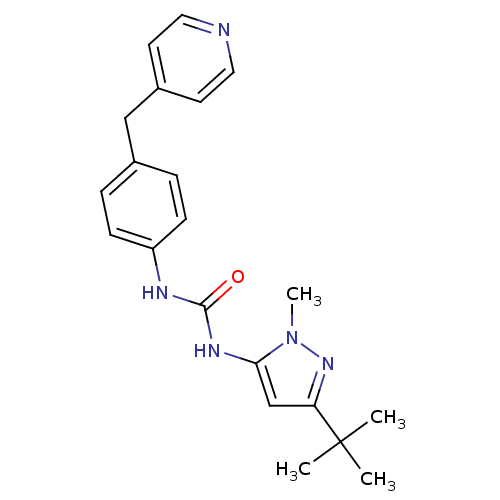
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dumas, J; Hatoum-Mokdad, H; Sibley, RN; Smith, RA; Scott, WJ; Khire, U; Lee, W; Wood, J; Wolanin, D; Cooley, J; Bankston, D; Redman, AM; Schoenleber, R; Caringal, Y; Gunn, D; Romero, R; Osterhout, M; Paulsen, H; Housley, TJ; Wilhelm, SM; Pirro, J; Chien, DS; Ranges, GE; Shrikhande, A; Muzsi, A; Bortolon, E; Wakefield, J; Gianpaolo Ostravage, C; Bhargava, A; Chau, T Synthesis and pharmacological characterization of a potent, orally active p38 kinase inhibitor. Bioorg Med Chem Lett12:1559-62 (2002) [PubMed]
Dumas, J; Hatoum-Mokdad, H; Sibley, RN; Smith, RA; Scott, WJ; Khire, U; Lee, W; Wood, J; Wolanin, D; Cooley, J; Bankston, D; Redman, AM; Schoenleber, R; Caringal, Y; Gunn, D; Romero, R; Osterhout, M; Paulsen, H; Housley, TJ; Wilhelm, SM; Pirro, J; Chien, DS; Ranges, GE; Shrikhande, A; Muzsi, A; Bortolon, E; Wakefield, J; Gianpaolo Ostravage, C; Bhargava, A; Chau, T Synthesis and pharmacological characterization of a potent, orally active p38 kinase inhibitor. Bioorg Med Chem Lett12:1559-62 (2002) [PubMed]