| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50125274 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_52064 |
|---|
| IC50 | 29000±n/a nM |
|---|
| Citation |  Blackie, JA; Bloomer, JC; Brown, MJ; Cheng, HY; Hammond, B; Hickey, DM; Ife, RJ; Leach, CA; Lewis, VA; Macphee, CH; Milliner, KJ; Moores, KE; Pinto, IL; Smith, SA; Stansfield, IG; Stanway, SJ; Taylor, MA; Theobald, CJ The identification of clinical candidate SB-480848: a potent inhibitor of lipoprotein-associated phospholipase A2. Bioorg Med Chem Lett13:1067-70 (2003) [PubMed] Blackie, JA; Bloomer, JC; Brown, MJ; Cheng, HY; Hammond, B; Hickey, DM; Ife, RJ; Leach, CA; Lewis, VA; Macphee, CH; Milliner, KJ; Moores, KE; Pinto, IL; Smith, SA; Stansfield, IG; Stanway, SJ; Taylor, MA; Theobald, CJ The identification of clinical candidate SB-480848: a potent inhibitor of lipoprotein-associated phospholipase A2. Bioorg Med Chem Lett13:1067-70 (2003) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50125274 |
|---|
| n/a |
|---|
| Name | BDBM50125274 |
|---|
| Synonyms: | 2-[5-Chloro-2-(4-fluoro-benzylsulfanyl)-4-oxo-4H-pyrimidin-1-yl]-N-(2-diethylamino-ethyl)-N-(4'-trifluoromethyl-biphenyl-4-ylmethyl)-acetamide | CHEMBL10769 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H33ClF4N4O2S |
|---|
| Mol. Mass. | 661.152 |
|---|
| SMILES | CCN(CC)CCN(Cc1ccc(cc1)-c1ccc(cc1)C(F)(F)F)C(=O)Cn1cc(Cl)c(=O)nc1SCc1ccc(F)cc1 |
|---|
| Structure | 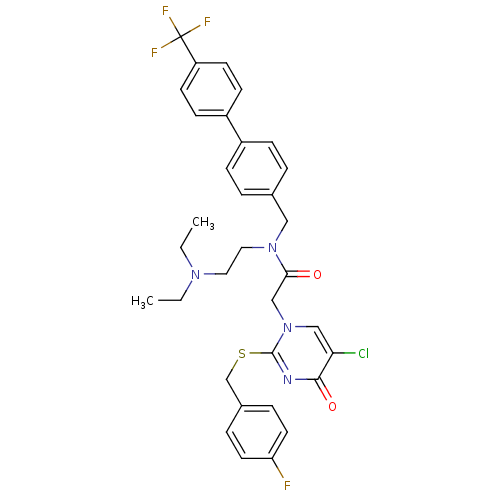
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Blackie, JA; Bloomer, JC; Brown, MJ; Cheng, HY; Hammond, B; Hickey, DM; Ife, RJ; Leach, CA; Lewis, VA; Macphee, CH; Milliner, KJ; Moores, KE; Pinto, IL; Smith, SA; Stansfield, IG; Stanway, SJ; Taylor, MA; Theobald, CJ The identification of clinical candidate SB-480848: a potent inhibitor of lipoprotein-associated phospholipase A2. Bioorg Med Chem Lett13:1067-70 (2003) [PubMed]
Blackie, JA; Bloomer, JC; Brown, MJ; Cheng, HY; Hammond, B; Hickey, DM; Ife, RJ; Leach, CA; Lewis, VA; Macphee, CH; Milliner, KJ; Moores, KE; Pinto, IL; Smith, SA; Stansfield, IG; Stanway, SJ; Taylor, MA; Theobald, CJ The identification of clinical candidate SB-480848: a potent inhibitor of lipoprotein-associated phospholipase A2. Bioorg Med Chem Lett13:1067-70 (2003) [PubMed]