| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A7 |
|---|
| Ligand | BDBM50584760 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2160655 (CHEMBL5045405) |
|---|
| IC50 | 10840±n/a nM |
|---|
| Citation |  Tu, D; Ning, J; Zou, L; Wang, P; Zhang, Y; Tian, X; Zhang, F; Zheng, J; Ge, G Unique Oxidative Metabolism of Bufalin Generates Two Reactive Metabolites That Strongly Inactivate Human Cytochrome P450 3A. J Med Chem65:4018-4029 (2022) [PubMed] Article Tu, D; Ning, J; Zou, L; Wang, P; Zhang, Y; Tian, X; Zhang, F; Zheng, J; Ge, G Unique Oxidative Metabolism of Bufalin Generates Two Reactive Metabolites That Strongly Inactivate Human Cytochrome P450 3A. J Med Chem65:4018-4029 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A7 |
|---|
| Name: | Cytochrome P450 3A7 |
|---|
| Synonyms: | 1.14.14.1 | CP3A7_HUMAN | CYP3A7 | CYPIIIA7 | Cytochrome P450 3A7 | Cytochrome P450-HFLA | P450HLp2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57485.39 |
|---|
| Organism: | Homo sapiens |
|---|
| Description: | ChEMBL_109496 |
|---|
| Residue: | 503 |
|---|
| Sequence: | MDLIPNLAVETWLLLAVSLILLYLYGTRTHGLFKKLGIPGPTPLPFLGNALSFRKGYWTF
DMECYKKYRKVWGIYDCQQPMLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKNAISI
AEDEEWKRIRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKHVFGAYS
MDVITSTSFGVSIDSLNNPQDPFVENTKKLLRFNPLDPFVLSIKVFPFLTPILEALNITV
FPRKVISFLTKSVKQIKEGRLKETQKHRVDFLQLMIDSQNSKDSETHKALSDLELMAQSI
IFIFAGYETTSSVLSFIIYELATHPDVQQKVQKEIDTVLPNKAPPTYDTVLQLEYLDMVV
NETLRLFPVAMRLERVCKKDVEINGMFIPKGVVVMIPSYVLHHDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALVNMKLALVRVLQNFSFKPCKETQIPLKLRFG
GLLLTEKPIVLKAESRDETVSGA
|
|
|
|---|
| BDBM50584760 |
|---|
| n/a |
|---|
| Name | BDBM50584760 |
|---|
| Synonyms: | CHEMBL2068968 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H32O4 |
|---|
| Mol. Mass. | 384.5085 |
|---|
| SMILES | [H][C@]12CC[C@]3([H])[C@]([H])(CC[C@]4(C)[C@H](CC[C@]34O)c3ccc(=O)oc3)[C@@]1(C)CCC(=O)C2 |
|---|
| Structure | 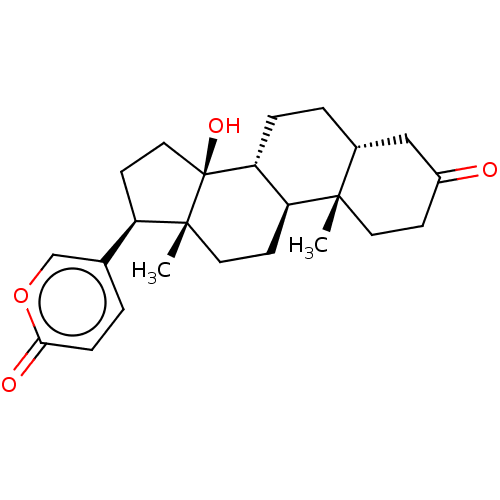
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tu, D; Ning, J; Zou, L; Wang, P; Zhang, Y; Tian, X; Zhang, F; Zheng, J; Ge, G Unique Oxidative Metabolism of Bufalin Generates Two Reactive Metabolites That Strongly Inactivate Human Cytochrome P450 3A. J Med Chem65:4018-4029 (2022) [PubMed] Article
Tu, D; Ning, J; Zou, L; Wang, P; Zhang, Y; Tian, X; Zhang, F; Zheng, J; Ge, G Unique Oxidative Metabolism of Bufalin Generates Two Reactive Metabolites That Strongly Inactivate Human Cytochrome P450 3A. J Med Chem65:4018-4029 (2022) [PubMed] Article