| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Pantetheinase |
|---|
| Ligand | BDBM394644 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2162813 (CHEMBL5047674) |
|---|
| IC50 | 4.1±n/a nM |
|---|
| Citation |  Casimiro-Garcia, A; Allais, C; Brennan, A; Choi, C; Dower, G; Farley, KA; Fleming, M; Flick, A; Frisbie, RK; Hall, J; Hepworth, D; Jones, H; Knafels, JD; Kortum, S; Lovering, FE; Mathias, JP; Mohan, S; Morgan, PM; Parng, C; Parris, K; Pullen, N; Schlerman, F; Stansfield, J; Strohbach, JW; Vajdos, FF; Vincent, F; Wang, H; Wang, X; Webster, R; Wright, SW Discovery of a Series of Pyrimidine Carboxamides as Inhibitors of Vanin-1. J Med Chem65:757-784 (2022) [PubMed] Article Casimiro-Garcia, A; Allais, C; Brennan, A; Choi, C; Dower, G; Farley, KA; Fleming, M; Flick, A; Frisbie, RK; Hall, J; Hepworth, D; Jones, H; Knafels, JD; Kortum, S; Lovering, FE; Mathias, JP; Mohan, S; Morgan, PM; Parng, C; Parris, K; Pullen, N; Schlerman, F; Stansfield, J; Strohbach, JW; Vajdos, FF; Vincent, F; Wang, H; Wang, X; Webster, R; Wright, SW Discovery of a Series of Pyrimidine Carboxamides as Inhibitors of Vanin-1. J Med Chem65:757-784 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Pantetheinase |
|---|
| Name: | Pantetheinase |
|---|
| Synonyms: | Pantetheine hydrolase | Tiff66 | VNN1 | VNN1_HUMAN | Vanin-1 | Vascular non-inflammatory molecule 1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 57002.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O95497 |
|---|
| Residue: | 513 |
|---|
| Sequence: | MTTQLPAYVAILLFYVSRASCQDTFTAAVYEHAAILPNATLTPVSREEALALMNRNLDIL
EGAITSAADQGAHIIVTPEDAIYGWNFNRDSLYPYLEDIPDPEVNWIPCNNRNRFGQTPV
QERLSCLAKNNSIYVVANIGDKKPCDTSDPQCPPDGRYQYNTDVVFDSQGKLVARYHKQN
LFMGENQFNVPKEPEIVTFNTTFGSFGIFTCFDILFHDPAVTLVKDFHVDTIVFPTAWMN
VLPHLSAVEFHSAWAMGMRVNFLASNIHYPSKKMTGSGIYAPNSSRAFHYDMKTEEGKLL
LSQLDSHPSHSAVVNWTSYASSIEALSSGNKEFKGTVFFDEFTFVKLTGVAGNYTVCQKD
LCCHLSYKMSENIPNEVYALGAFDGLHTVEGRYYLQICTLLKCKTTNLNTCGDSAETAST
RFEMFSLSGTFGTQYVFPEVLLSENQLAPGEFQVSTDGRLFSLKPTSGPVLTVTLFGRLY
EKDWASNASSGLTAQARIIMLIVIAPIVCSLSW
|
|
|
|---|
| BDBM394644 |
|---|
| n/a |
|---|
| Name | BDBM394644 |
|---|
| Synonyms: | 3-[(2-{[1-(pyrimidin-5-yl)cyclopropyl]amino}pyrimidin-5-yl)carbonyl]benzonitrile | US10308615, Example 1 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H14N6O |
|---|
| Mol. Mass. | 342.3541 |
|---|
| SMILES | O=C(c1cnc(NC2(CC2)c2cncnc2)nc1)c1cccc(c1)C#N |
|---|
| Structure | 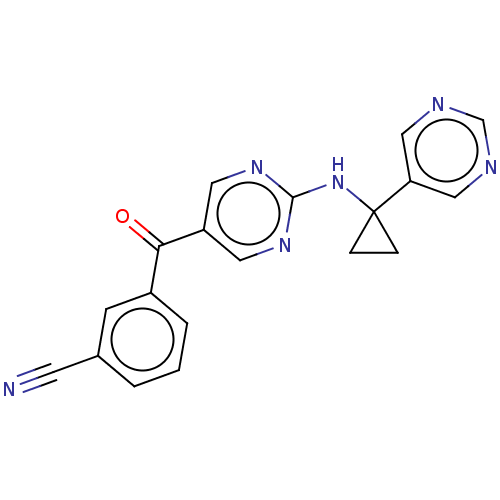
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Casimiro-Garcia, A; Allais, C; Brennan, A; Choi, C; Dower, G; Farley, KA; Fleming, M; Flick, A; Frisbie, RK; Hall, J; Hepworth, D; Jones, H; Knafels, JD; Kortum, S; Lovering, FE; Mathias, JP; Mohan, S; Morgan, PM; Parng, C; Parris, K; Pullen, N; Schlerman, F; Stansfield, J; Strohbach, JW; Vajdos, FF; Vincent, F; Wang, H; Wang, X; Webster, R; Wright, SW Discovery of a Series of Pyrimidine Carboxamides as Inhibitors of Vanin-1. J Med Chem65:757-784 (2022) [PubMed] Article
Casimiro-Garcia, A; Allais, C; Brennan, A; Choi, C; Dower, G; Farley, KA; Fleming, M; Flick, A; Frisbie, RK; Hall, J; Hepworth, D; Jones, H; Knafels, JD; Kortum, S; Lovering, FE; Mathias, JP; Mohan, S; Morgan, PM; Parng, C; Parris, K; Pullen, N; Schlerman, F; Stansfield, J; Strohbach, JW; Vajdos, FF; Vincent, F; Wang, H; Wang, X; Webster, R; Wright, SW Discovery of a Series of Pyrimidine Carboxamides as Inhibitors of Vanin-1. J Med Chem65:757-784 (2022) [PubMed] Article