| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Ligand | BDBM50342580 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_744966 (CHEMBL1771981) |
|---|
| IC50 | 1400±n/a nM |
|---|
| Citation |  Klöck, C; Jin, X; Choi, K; Khosla, C; Madrid, PB; Spencer, A; Raimundo, BC; Boardman, P; Lanza, G; Griffin, JH Acylideneoxoindoles: a new class of reversible inhibitors of human transglutaminase 2. Bioorg Med Chem Lett21:2692-6 (2011) [PubMed] Article Klöck, C; Jin, X; Choi, K; Khosla, C; Madrid, PB; Spencer, A; Raimundo, BC; Boardman, P; Lanza, G; Griffin, JH Acylideneoxoindoles: a new class of reversible inhibitors of human transglutaminase 2. Bioorg Med Chem Lett21:2692-6 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Name: | Protein-glutamine gamma-glutamyltransferase 2 |
|---|
| Synonyms: | Protein-glutamine gamma-glutamyltransferase | Protein-glutamine gamma-glutamyltransferase 2 (TG2) | TGM2 | TGM2_HUMAN | Tissue transglutaminase | Tissue transglutaminase (TG2) | Transglutaminase 2 (TGM2) | Transglutaminase C | Transglutaminase-2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 77309.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21980 |
|---|
| Residue: | 687 |
|---|
| Sequence: | MAEELVLERCDLELETNGRDHHTADLCREKLVVRRGQPFWLTLHFEGRNYEASVDSLTFS
VVTGPAPSQEAGTKARFPLRDAVEEGDWTATVVDQQDCTLSLQLTTPANAPIGLYRLSLE
ASTGYQGSSFVLGHFILLFNAWCPADAVYLDSEEERQEYVLTQQGFIYQGSAKFIKNIPW
NFGQFEDGILDICLILLDVNPKFLKNAGRDCSRRSSPVYVGRVVSGMVNCNDDQGVLLGR
WDNNYGDGVSPMSWIGSVDILRRWKNHGCQRVKYGQCWVFAAVACTVLRCLGIPTRVVTN
YNSAHDQNSNLLIEYFRNEFGEIQGDKSEMIWNFHCWVESWMTRPDLQPGYEGWQALDPT
PQEKSEGTYCCGPVPVRAIKEGDLSTKYDAPFVFAEVNADVVDWIQQDDGSVHKSINRSL
IVGLKISTKSVGRDEREDITHTYKYPEGSSEEREAFTRANHLNKLAEKEETGMAMRIRVG
QSMNMGSDFDVFAHITNNTAEEYVCRLLLCARTVSYNGILGPECGTKYLLNLNLEPFSEK
SVPLCILYEKYRDCLTESNLIKVRALLVEPVINSYLLAERDLYLENPEIKIRILGEPKQK
RKLVAEVSLQNPLPVALEGCTFTVEGAGLTEEQKTVEIPDPVEAGEEVKVRMDLLPLHMG
LHKLVVNFESDKLKAVKGFRNVIIGPA
|
|
|
|---|
| BDBM50342580 |
|---|
| n/a |
|---|
| Name | BDBM50342580 |
|---|
| Synonyms: | (E)-4-chloro-3-(2-(6-methoxypyridin-3-yl)-2-oxoethylidene)indolin-2-one | CHEMBL1770517 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H11ClN2O3 |
|---|
| Mol. Mass. | 314.723 |
|---|
| SMILES | COc1ccncc1C(=O)\C=C1\C(=O)Nc2cccc(Cl)c12 |
|---|
| Structure | 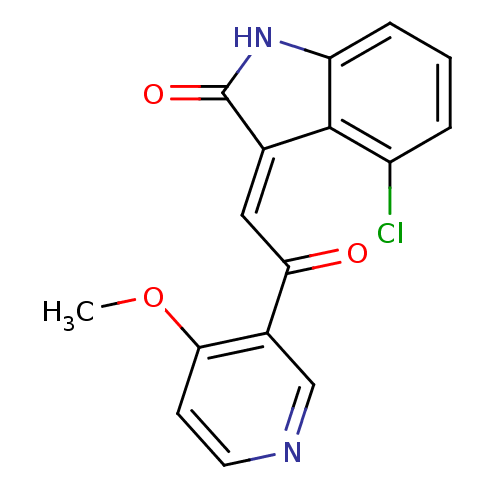
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Klöck, C; Jin, X; Choi, K; Khosla, C; Madrid, PB; Spencer, A; Raimundo, BC; Boardman, P; Lanza, G; Griffin, JH Acylideneoxoindoles: a new class of reversible inhibitors of human transglutaminase 2. Bioorg Med Chem Lett21:2692-6 (2011) [PubMed] Article
Klöck, C; Jin, X; Choi, K; Khosla, C; Madrid, PB; Spencer, A; Raimundo, BC; Boardman, P; Lanza, G; Griffin, JH Acylideneoxoindoles: a new class of reversible inhibitors of human transglutaminase 2. Bioorg Med Chem Lett21:2692-6 (2011) [PubMed] Article