| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Substance-K receptor |
|---|
| Ligand | BDBM50279775 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_209035 |
|---|
| Ki | 0.600000±n/a nM |
|---|
| Citation |  Bernstein, PR; Aharony, D; Albert, JS; Andisik, D; Barthlow, HG; Bialecki, R; Davenport, T; Dedinas, RF; Dembofsky, BT; Koether, G; Kosmider, BJ; Kirkland, K; Ohnmacht, CJ; Potts, W; Rumsey, WL; Shen, L; Shenvi, A; Sherwood, S; Stollman, D; Russell, K Discovery of novel, orally active dual NK1/NK2 antagonists. Bioorg Med Chem Lett11:2769-73 (2001) [PubMed] Bernstein, PR; Aharony, D; Albert, JS; Andisik, D; Barthlow, HG; Bialecki, R; Davenport, T; Dedinas, RF; Dembofsky, BT; Koether, G; Kosmider, BJ; Kirkland, K; Ohnmacht, CJ; Potts, W; Rumsey, WL; Shen, L; Shenvi, A; Sherwood, S; Stollman, D; Russell, K Discovery of novel, orally active dual NK1/NK2 antagonists. Bioorg Med Chem Lett11:2769-73 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Substance-K receptor |
|---|
| Name: | Substance-K receptor |
|---|
| Synonyms: | NK-2 receptor | NK-2R | NK2R | NK2R_HUMAN | NKNAR | Neurokinin 2 receptor | Neurokinin A receptor | Neurokinin NK2 | Neurokinin-2 (NK-2) | Neuromedin-2 receptor (NK-2R) | SKR | TAC2R | TACR2 | Tachykinin receptor 2 | Tachykinin receptor 2 (NK2) | hnk-3 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 44455.78 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21452 |
|---|
| Residue: | 398 |
|---|
| Sequence: | MGTCDIVTEANISSGPESNTTGITAFSMPSWQLALWATAYLALVLVAVTGNAIVIWIILA
HRRMRTVTNYFIVNLALADLCMAAFNAAFNFVYASHNIWYFGRAFCYFQNLFPITAMFVS
IYSMTAIAADRYMAIVHPFQPRLSAPSTKAVIAGIWLVALALASPQCFYSTVTMDQGATK
CVVAWPEDSGGKTLLLYHLVVIALIYFLPLAVMFVAYSVIGLTLWRRAVPGHQAHGANLR
HLQAMKKFVKTMVLVVLTFAICWLPYHLYFILGSFQEDIYCHKFIQQVYLALFWLAMSST
MYNPIIYCCLNHRFRSGFRLAFRCCPWVTPTKEDKLELTPTTSLSTRVNRCHTKETLFMA
GDTAPSEATSGEAGRPQDGSGLWFGYGLLAPTKTHVEI
|
|
|
|---|
| BDBM50279775 |
|---|
| n/a |
|---|
| Name | BDBM50279775 |
|---|
| Synonyms: | (S)-3-Cyano-naphthalene-1-carboxylic acid {2-(3,4-dichloro-phenyl)-4-[4-(2-methanesulfinyl-phenyl)-piperidin-1-yl]-butyl}-methyl-amide | 3-Cyano-naphthalene-1-carboxylic acid ((S)-2-(3,4-dichloro-phenyl)-4-{4-[2-((S)-methanesulfinyl)-phenyl]-piperidin-1-yl}-butyl)-methyl-amide | CHEMBL129683 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C35H35Cl2N3O2S |
|---|
| Mol. Mass. | 632.642 |
|---|
| SMILES | CN(C[C@@H](CCN1CCC(CC1)c1ccccc1[S@](C)=O)c1ccc(Cl)c(Cl)c1)C(=O)c1cc(cc2ccccc12)C#N |
|---|
| Structure | 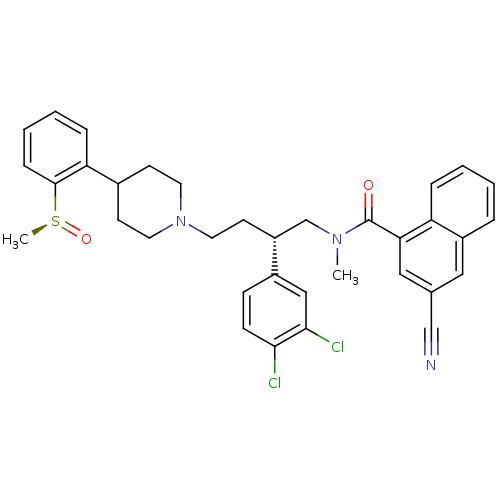
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bernstein, PR; Aharony, D; Albert, JS; Andisik, D; Barthlow, HG; Bialecki, R; Davenport, T; Dedinas, RF; Dembofsky, BT; Koether, G; Kosmider, BJ; Kirkland, K; Ohnmacht, CJ; Potts, W; Rumsey, WL; Shen, L; Shenvi, A; Sherwood, S; Stollman, D; Russell, K Discovery of novel, orally active dual NK1/NK2 antagonists. Bioorg Med Chem Lett11:2769-73 (2001) [PubMed]
Bernstein, PR; Aharony, D; Albert, JS; Andisik, D; Barthlow, HG; Bialecki, R; Davenport, T; Dedinas, RF; Dembofsky, BT; Koether, G; Kosmider, BJ; Kirkland, K; Ohnmacht, CJ; Potts, W; Rumsey, WL; Shen, L; Shenvi, A; Sherwood, S; Stollman, D; Russell, K Discovery of novel, orally active dual NK1/NK2 antagonists. Bioorg Med Chem Lett11:2769-73 (2001) [PubMed]