| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Tumor susceptibility gene 101 protein |
|---|
| Ligand | BDBM50230455 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_461905 (CHEMBL944752) |
|---|
| IC50 | 16000±n/a nM |
|---|
| Citation |  Liu, F; Stephen, AG; Fisher, RJ; Burke, TR Protected aminooxyprolines for expedited library synthesis: application to Tsg101-directed proline-oxime containing peptides. Bioorg Med Chem Lett18:1096-101 (2008) [PubMed] Article Liu, F; Stephen, AG; Fisher, RJ; Burke, TR Protected aminooxyprolines for expedited library synthesis: application to Tsg101-directed proline-oxime containing peptides. Bioorg Med Chem Lett18:1096-101 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Tumor susceptibility gene 101 protein |
|---|
| Name: | Tumor susceptibility gene 101 protein |
|---|
| Synonyms: | TS101_HUMAN | TSG101 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 43943.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_800321 |
|---|
| Residue: | 390 |
|---|
| Sequence: | MAVSESQLKKMVSKYKYRDLTVRETVNVITLYKDLKPVLDSYVFNDGSSRELMNLTGTIP
VPYRGNTYNIPICLWLLDTYPYNPPICFVKPTSSMTIKTGKHVDANGKIYLPYLHEWKHP
QSDLLGLIQVMIVVFGDEPPVFSRPISASYPPYQATGPPNTSYMPGMPGGISPYPSGYPP
NPSGYPGCPYPPGGPYPATTSSQYPSQPPVTTVGPSRDGTISEDTIRASLISAVSDKLRW
RMKEEMDRAQAELNALKRTEEDLKKGHQKLEEMVTRLDQEVAEVDKNIELLKKKDEELSS
ALEKMENQSENNDIDEVIIPTAPLYKQILNLYAEENAIEDTIFYLGEALRRGVIDLDVFL
KHVRLLSRKQFQLRALMQKARKTAGLSDLY
|
|
|
|---|
| BDBM50230455 |
|---|
| n/a |
|---|
| Name | BDBM50230455 |
|---|
| Synonyms: | (4S)-4-{[(1S)-1-carbamoyl-3-carboxypropyl]carbamoyl}-4-{[(2S,3S)-1-({1-[(2S)-2-[(2S,3R)-2-{[(2S)-1-[(2S)-4-carboxy-2-{[(2S)-1-{5-[({3',6'-dihydroxy-3-oxo-3H-spiro[2-benzofuran-1,9'-xanthene]-6-yl}carbamothioyl)amino]pentanoyl}pyrrolidin-2-yl]formamido}butanoyl]pyrrolidin-2-yl]formamido}-3-hydroxybutanamido]propanoyl]piperidin-2-yl}carbonyl)-3-{[(Z)-(3-carboxypropylidene)amino]oxy}pyrrolidin-2-yl]formamido}butanoic acid | CHEMBL411417 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C73H91N13O25S |
|---|
| Mol. Mass. | 1582.641 |
|---|
| SMILES | C[C@@H](O)[C@H](NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCC(O)=O)NC(=O)[C@@H]1CCCN1C(=O)CCCCNC(=S)Nc1ccc2C(=O)OC3(c2c1)c1ccc(O)cc1Oc1cc(O)ccc31)C(=O)N[C@@H](C)C(=O)N1CCCCC1C(=O)N1CC[C@H](O\N=C/CCC(O)=O)[C@H]1C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(N)=O |
|---|
| Structure | 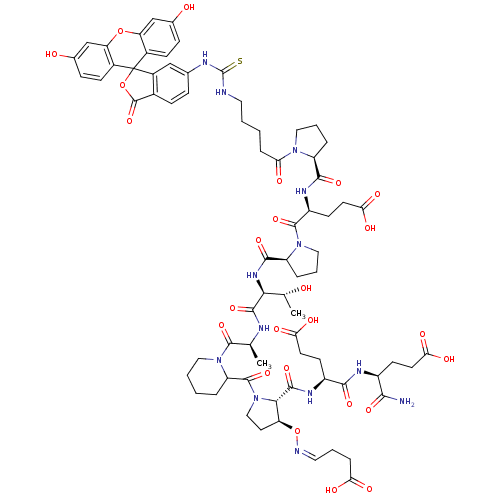
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liu, F; Stephen, AG; Fisher, RJ; Burke, TR Protected aminooxyprolines for expedited library synthesis: application to Tsg101-directed proline-oxime containing peptides. Bioorg Med Chem Lett18:1096-101 (2008) [PubMed] Article
Liu, F; Stephen, AG; Fisher, RJ; Burke, TR Protected aminooxyprolines for expedited library synthesis: application to Tsg101-directed proline-oxime containing peptides. Bioorg Med Chem Lett18:1096-101 (2008) [PubMed] Article