| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50206351 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_564940 (CHEMBL956718) |
|---|
| Ki | 6.51±n/a nM |
|---|
| Citation |  Zajdel, P; Subra, G; Verdie, P; Bojarski, AJ; Duszynska, B; Basista, K; Obniska, J; Martinez, J; Pawlowski, M The influence of an ethylene spacer on the 5-HT(1A) and 5-HT(2A) receptor affinity of arylpiperazine derivatives of amides with N-acylated amino acids and 3-differently substituted pyrrolidine-2,5-diones. Eur J Med Chem44:800-8 (2009) [PubMed] Article Zajdel, P; Subra, G; Verdie, P; Bojarski, AJ; Duszynska, B; Basista, K; Obniska, J; Martinez, J; Pawlowski, M The influence of an ethylene spacer on the 5-HT(1A) and 5-HT(2A) receptor affinity of arylpiperazine derivatives of amides with N-acylated amino acids and 3-differently substituted pyrrolidine-2,5-diones. Eur J Med Chem44:800-8 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM50206351 |
|---|
| n/a |
|---|
| Name | BDBM50206351 |
|---|
| Synonyms: | (S)-N-(1-(4-(4-(3-chlorophenyl)piperazin-1-yl)butyl)-2,5-dioxopyrrolidin-3-yl)cyclohexanecarboxamide | CHEMBL408589 | N-((S)-1-{4-[4-(3-chlorophenyl)piperazin-1-yl]butyl}pyrrolidin-2,5-dion-3-yl)cyclohexanecarboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H35ClN4O3 |
|---|
| Mol. Mass. | 475.023 |
|---|
| SMILES | Clc1cccc(c1)N1CCN(CCCCN2C(=O)C[C@H](NC(=O)C3CCCCC3)C2=O)CC1 |r| |
|---|
| Structure | 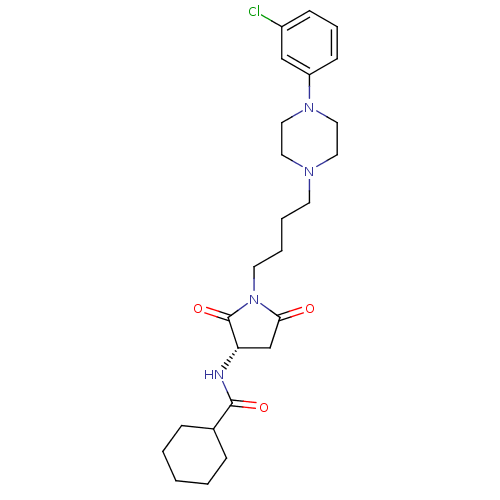
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zajdel, P; Subra, G; Verdie, P; Bojarski, AJ; Duszynska, B; Basista, K; Obniska, J; Martinez, J; Pawlowski, M The influence of an ethylene spacer on the 5-HT(1A) and 5-HT(2A) receptor affinity of arylpiperazine derivatives of amides with N-acylated amino acids and 3-differently substituted pyrrolidine-2,5-diones. Eur J Med Chem44:800-8 (2009) [PubMed] Article
Zajdel, P; Subra, G; Verdie, P; Bojarski, AJ; Duszynska, B; Basista, K; Obniska, J; Martinez, J; Pawlowski, M The influence of an ethylene spacer on the 5-HT(1A) and 5-HT(2A) receptor affinity of arylpiperazine derivatives of amides with N-acylated amino acids and 3-differently substituted pyrrolidine-2,5-diones. Eur J Med Chem44:800-8 (2009) [PubMed] Article