| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor X |
|---|
| Ligand | BDBM50270656 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_603964 (CHEMBL1043317) |
|---|
| IC50 | 4.8±n/a nM |
|---|
| Citation |  Fujimoto, T; Tobisu, M; Konishi, N; Kawamura, M; Tada, N; Takagi, T; Kubo, K Synthesis and biological evaluation of the metabolites of 2-(1-{3-[(6-chloronaphthalen-2-yl)sulfonyl]propanoyl}piperidin-4-yl)-5-methyl-1,2-dihydro-3H-imidazo[1,5-c]imidazol-3-one. Bioorg Med Chem17:7993-8002 (2009) [PubMed] Article Fujimoto, T; Tobisu, M; Konishi, N; Kawamura, M; Tada, N; Takagi, T; Kubo, K Synthesis and biological evaluation of the metabolites of 2-(1-{3-[(6-chloronaphthalen-2-yl)sulfonyl]propanoyl}piperidin-4-yl)-5-methyl-1,2-dihydro-3H-imidazo[1,5-c]imidazol-3-one. Bioorg Med Chem17:7993-8002 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor X |
|---|
| Name: | Coagulation factor X |
|---|
| Synonyms: | Activated coagulation factor X (FXa) | Activated factor Xa heavy chain | Coagulation factor X precursor | Coagulation factor Xa | F10 | FA10_HUMAN | Factor X heavy chain | Factor X light chain | Factor Xa | Stuart factor | Stuart-Prower factor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 54726.60 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 488 |
|---|
| Sequence: | MGRPLHLVLLSASLAGLLLLGESLFIRREQANNILARVTRANSFLEEMKKGHLERECMEE
TCSYEEAREVFEDSDKTNEFWNKYKDGDQCETSPCQNQGKCKDGLGEYTCTCLEGFEGKN
CELFTRKLCSLDNGDCDQFCHEEQNSVVCSCARGYTLADNGKACIPTGPYPCGKQTLERR
KRSVAQATSSSGEAPDSITWKPYDAADLDPTENPFDLLDFNQTQPERGDNNLTRIVGGQE
CKDGECPWQALLINEENEGFCGGTILSEFYILTAAHCLYQAKRFKVRVGDRNTEQEEGGE
AVHEVEVVIKHNRFTKETYDFDIAVLRLKTPITFRMNVAPACLPERDWAESTLMTQKTGI
VSGFGRTHEKGRQSTRLKMLEVPYVDRNSCKLSSSFIITQNMFCAGYDTKQEDACQGDSG
GPHVTRFKDTYFVTGIVSWGEGCARKGKYGIYTKVTAFLKWIDRSMKTRGLPKAKSHAPE
VITSSPLK
|
|
|
|---|
| BDBM50270656 |
|---|
| n/a |
|---|
| Name | BDBM50270656 |
|---|
| Synonyms: | 2-(1-(3-(6-chloronaphthalen-2-ylsulfonyl)propanoyl)piperidin-4-yl)-5-methyl-1,2-dihydroimidazo[1,5-e]imidazol-3-one | 2-(1-(3-(6-chloronaphthalen-2-ylsulfonyl)propanoyl)piperidin-4-yl)-5-methyl-1H-imidazo[1,5-c]imidazol-3(2H)-one | CHEMBL487105 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H25ClN4O4S |
|---|
| Mol. Mass. | 500.998 |
|---|
| SMILES | Cc1ncc2CN(C3CCN(CC3)C(=O)CCS(=O)(=O)c3ccc4cc(Cl)ccc4c3)C(=O)n12 |
|---|
| Structure | 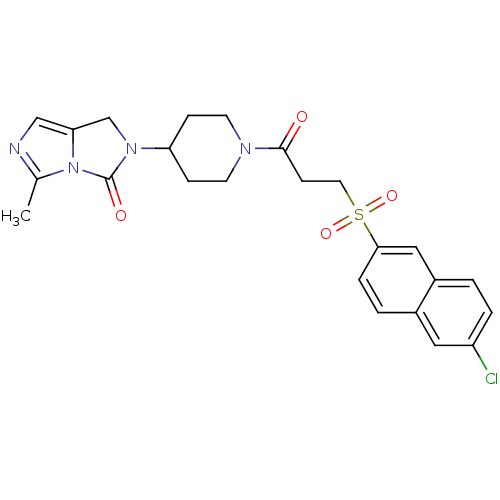
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fujimoto, T; Tobisu, M; Konishi, N; Kawamura, M; Tada, N; Takagi, T; Kubo, K Synthesis and biological evaluation of the metabolites of 2-(1-{3-[(6-chloronaphthalen-2-yl)sulfonyl]propanoyl}piperidin-4-yl)-5-methyl-1,2-dihydro-3H-imidazo[1,5-c]imidazol-3-one. Bioorg Med Chem17:7993-8002 (2009) [PubMed] Article
Fujimoto, T; Tobisu, M; Konishi, N; Kawamura, M; Tada, N; Takagi, T; Kubo, K Synthesis and biological evaluation of the metabolites of 2-(1-{3-[(6-chloronaphthalen-2-yl)sulfonyl]propanoyl}piperidin-4-yl)-5-methyl-1,2-dihydro-3H-imidazo[1,5-c]imidazol-3-one. Bioorg Med Chem17:7993-8002 (2009) [PubMed] Article