| Reaction Details |
|---|
 | Report a problem with these data |
| Target | RAC-gamma serine/threonine-protein kinase |
|---|
| Ligand | BDBM50427345 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_941087 (CHEMBL2330884) |
|---|
| IC50 | 24±n/a nM |
|---|
| Citation |  Addie, M; Ballard, P; Buttar, D; Crafter, C; Currie, G; Davies, BR; Debreczeni, J; Dry, H; Dudley, P; Greenwood, R; Johnson, PD; Kettle, JG; Lane, C; Lamont, G; Leach, A; Luke, RW; Morris, J; Ogilvie, D; Page, K; Pass, M; Pearson, S; Ruston, L Discovery of 4-amino-N-[(1S)-1-(4-chlorophenyl)-3-hydroxypropyl]-1-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)piperidine-4-carboxamide (AZD5363), an orally bioavailable, potent inhibitor of Akt kinases. J Med Chem56:2059-73 (2013) [PubMed] Article Addie, M; Ballard, P; Buttar, D; Crafter, C; Currie, G; Davies, BR; Debreczeni, J; Dry, H; Dudley, P; Greenwood, R; Johnson, PD; Kettle, JG; Lane, C; Lamont, G; Leach, A; Luke, RW; Morris, J; Ogilvie, D; Page, K; Pass, M; Pearson, S; Ruston, L Discovery of 4-amino-N-[(1S)-1-(4-chlorophenyl)-3-hydroxypropyl]-1-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)piperidine-4-carboxamide (AZD5363), an orally bioavailable, potent inhibitor of Akt kinases. J Med Chem56:2059-73 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| RAC-gamma serine/threonine-protein kinase |
|---|
| Name: | RAC-gamma serine/threonine-protein kinase |
|---|
| Synonyms: | AKT3 | AKT3_HUMAN | PKB gamma | PKBG | Protein kinase Akt-3 | Protein kinase B (Akt 3) | Protein kinase B, gamma | RAC-PK-gamma | RAC-gamma serine/threonine-protein kinase | STK-2 | Serine/threonine-protein kinase AKT | Serine/threonine-protein kinase AKT3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55769.36 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 479 |
|---|
| Sequence: | MSDVTIVKEGWVQKRGEYIKNWRPRYFLLKTDGSFIGYKEKPQDVDLPYPLNNFSVAKCQ
LMKTERPKPNTFIIRCLQWTTVIERTFHVDTPEEREEWTEAIQAVADRLQRQEEERMNCS
PTSQIDNIGEEEMDASTTHHKRKTMNDFDYLKLLGKGTFGKVILVREKASGKYYAMKILK
KEVIIAKDEVAHTLTESRVLKNTRHPFLTSLKYSFQTKDRLCFVMEYVNGGELFFHLSRE
RVFSEDRTRFYGAEIVSALDYLHSGKIVYRDLKLENLMLDKDGHIKITDFGLCKEGITDA
ATMKTFCGTPEYLAPEVLEDNDYGRAVDWWGLGVVMYEMMCGRLPFYNQDHEKLFELILM
EDIKFPRTLSSDAKSLLSGLLIKDPNKRLGGGPDDAKEIMRHSFFSGVNWQDVYDKKLVP
PFKPQVTSETDTRYFDEEFTAQTITITPPEKYDEDGMDCMDNERRPHFPQFSYSASGRE
|
|
|
|---|
| BDBM50427345 |
|---|
| n/a |
|---|
| Name | BDBM50427345 |
|---|
| Synonyms: | CHEMBL2325983 | US10654855, Example 4 | US11236095, Example 4 | US9492453, 4 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H25ClN6O |
|---|
| Mol. Mass. | 424.927 |
|---|
| SMILES | NC1(CCN(CC1)c1ncnc2[nH]ccc12)C(=O)NC(C1CC1)c1ccc(Cl)cc1 |
|---|
| Structure | 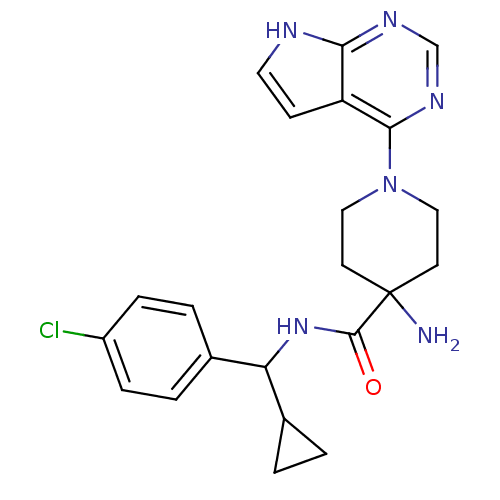
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Addie, M; Ballard, P; Buttar, D; Crafter, C; Currie, G; Davies, BR; Debreczeni, J; Dry, H; Dudley, P; Greenwood, R; Johnson, PD; Kettle, JG; Lane, C; Lamont, G; Leach, A; Luke, RW; Morris, J; Ogilvie, D; Page, K; Pass, M; Pearson, S; Ruston, L Discovery of 4-amino-N-[(1S)-1-(4-chlorophenyl)-3-hydroxypropyl]-1-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)piperidine-4-carboxamide (AZD5363), an orally bioavailable, potent inhibitor of Akt kinases. J Med Chem56:2059-73 (2013) [PubMed] Article
Addie, M; Ballard, P; Buttar, D; Crafter, C; Currie, G; Davies, BR; Debreczeni, J; Dry, H; Dudley, P; Greenwood, R; Johnson, PD; Kettle, JG; Lane, C; Lamont, G; Leach, A; Luke, RW; Morris, J; Ogilvie, D; Page, K; Pass, M; Pearson, S; Ruston, L Discovery of 4-amino-N-[(1S)-1-(4-chlorophenyl)-3-hydroxypropyl]-1-(7H-pyrrolo[2,3-d]pyrimidin-4-yl)piperidine-4-carboxamide (AZD5363), an orally bioavailable, potent inhibitor of Akt kinases. J Med Chem56:2059-73 (2013) [PubMed] Article