

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Prothrombin | ||
| Ligand | BDBM254896 | ||
| Substrate/Competitor | BDBM254912 | ||
| Meas. Tech. | Inhibition Assay | ||
| pH | 7.4±n/a | ||
| Temperature | 295.15±n/a K | ||
| IC50 | 0.30±n/a nM | ||
| Comments | extracted | ||
| Citation |  Allerheiligen, S; Buchmüller, A; Engel, K; Gerdes, C; Gericke, KM; Gerisch, M; Heitmeier, S; Hillisch, A; Kinzel, T; Lienau, P; Riedl, B; Röhrig, S; Schmidt, MV; Strassburger, J; Tersteegen, A Substituted benzoxazoles US Patent US9493472 Publication Date 11/15/2016 Allerheiligen, S; Buchmüller, A; Engel, K; Gerdes, C; Gericke, KM; Gerisch, M; Heitmeier, S; Hillisch, A; Kinzel, T; Lienau, P; Riedl, B; Röhrig, S; Schmidt, MV; Strassburger, J; Tersteegen, A Substituted benzoxazoles US Patent US9493472 Publication Date 11/15/2016 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Prothrombin | |||
| Name: | Prothrombin | ||
| Synonyms: | Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain | ||
| Type: | Protein | ||
| Mol. Mass.: | 70029.57 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P00734 | ||
| Residue: | 622 | ||
| Sequence: |
| ||
| BDBM254896 | |||
| BDBM254912 | |||
| Name | BDBM254896 | ||
| Synonyms: | US9493472, 18 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C26H29ClFN3O5 | ||
| Mol. Mass. | 517.977 | ||
| SMILES | COc1cc(cc2nc(NC(CF)c3cccc(Cl)c3)oc12)C(=O)N1C[C@H](C)OC[C@H]1C1CC(O)C1 |r,wU:26.29,wD:30.34,(-3.32,5.32,;-1.98,4.55,;-1.98,3.01,;-3.32,2.24,;-3.32,.7,;-1.98,-.07,;-.65,.7,;.82,.23,;1.72,1.47,;3.26,1.47,;4.03,.14,;3.26,-1.19,;4.03,-2.53,;5.57,.14,;6.34,1.47,;7.88,1.47,;8.65,.14,;7.88,-1.19,;8.65,-2.53,;6.34,-1.19,;.82,2.72,;-.65,2.24,;-4.65,-.07,;-4.65,-1.61,;-5.98,.7,;-5.98,2.24,;-7.32,3.01,;-7.32,4.55,;-8.65,2.24,;-8.65,.7,;-7.32,-.07,;-7.32,-1.61,;-8.41,-2.69,;-7.32,-3.78,;-7.32,-5.32,;-6.23,-2.69,)| | ||
| Structure | 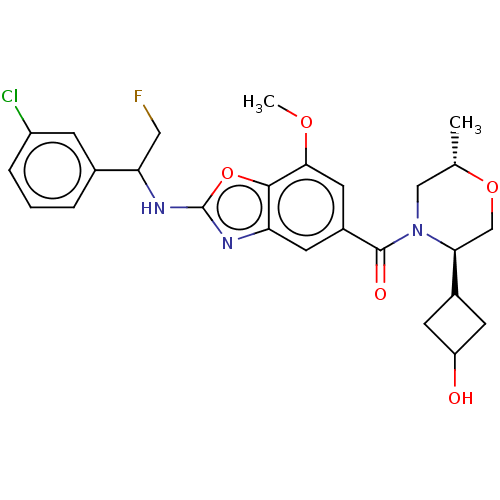
| ||