| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fibroblast growth factor receptor 4 |
|---|
| Ligand | BDBM443222 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition Assay |
|---|
| IC50 | 2.00±n/a nM |
|---|
| Citation |  Chen, L; Guan, D; Bai, H; Gou, J; Zhao, W; Wang, Z; Ling, L; Ma, Y Pyrimidine derivative, method for preparing same and use thereof in medicine US Patent US10654836 Publication Date 5/19/2020 Chen, L; Guan, D; Bai, H; Gou, J; Zhao, W; Wang, Z; Ling, L; Ma, Y Pyrimidine derivative, method for preparing same and use thereof in medicine US Patent US10654836 Publication Date 5/19/2020 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Fibroblast growth factor receptor 4 |
|---|
| Name: | Fibroblast growth factor receptor 4 |
|---|
| Synonyms: | CD_antigen: CD334 FGFR4 | FGFR-4 | FGFR4 | FGFR4_HUMAN | Fibroblast growth factor receptor | Fibroblast growth factor receptor 4 (FGFR4) | JK2 | JTK2 | TKF |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 87957.45 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P22455 |
|---|
| Residue: | 802 |
|---|
| Sequence: | MRLLLALLGVLLSVPGPPVLSLEASEEVELEPCLAPSLEQQEQELTVALGQPVRLCCGRA
ERGGHWYKEGSRLAPAGRVRGWRGRLEIASFLPEDAGRYLCLARGSMIVLQNLTLITGDS
LTSSNDDEDPKSHRDPSNRHSYPQQAPYWTHPQRMEKKLHAVPAGNTVKFRCPAAGNPTP
TIRWLKDGQAFHGENRIGGIRLRHQHWSLVMESVVPSDRGTYTCLVENAVGSIRYNYLLD
VLERSPHRPILQAGLPANTTAVVGSDVELLCKVYSDAQPHIQWLKHIVINGSSFGADGFP
YVQVLKTADINSSEVEVLYLRNVSAEDAGEYTCLAGNSIGLSYQSAWLTVLPEEDPTWTA
AAPEARYTDIILYASGSLALAVLLLLAGLYRGQALHGRHPRPPATVQKLSRFPLARQFSL
ESGSSGKSSSSLVRGVRLSSSGPALLAGLVSLDLPLDPLWEFPRDRLVLGKPLGEGCFGQ
VVRAEAFGMDPARPDQASTVAVKMLKDNASDKDLADLVSEMEVMKLIGRHKNIINLLGVC
TQEGPLYVIVECAAKGNLREFLRARRPPGPDLSPDGPRSSEGPLSFPVLVSCAYQVARGM
QYLESRKCIHRDLAARNVLVTEDNVMKIADFGLARGVHHIDYYKKTSNGRLPVKWMAPEA
LFDRVYTHQSDVWSFGILLWEIFTLGGSPYPGIPVEELFSLLREGHRMDRPPHCPPELYG
LMRECWHAAPSQRPTFKQLVEALDKVLLAVSEEYLDLRLTFGPYSPSGGDASSTCSSSDS
VFSHDPLPLGSSSFPFGSGVQT
|
|
|
|---|
| BDBM443222 |
|---|
| n/a |
|---|
| Name | BDBM443222 |
|---|
| Synonyms: | N-(2-((6-(3-(2,6-Dichloro-3,5-dimethoxyphenyl)-1-methylureido)pyrimidin-4-yl)amino)-5-(2,8-diazaspiro[4.5]decan-2-yl)phenyl)acrylamide | US10654836, Example 3 | US11001572, Example 3 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H36Cl2N8O4 |
|---|
| Mol. Mass. | 655.575 |
|---|
| SMILES | COc1cc(OC)c(Cl)c(NC(=O)N(C)c2cc(Nc3ccc(cc3NC(=O)C=C)N3CCC4(C3)CCNCC4)ncn2)c1Cl |
|---|
| Structure | 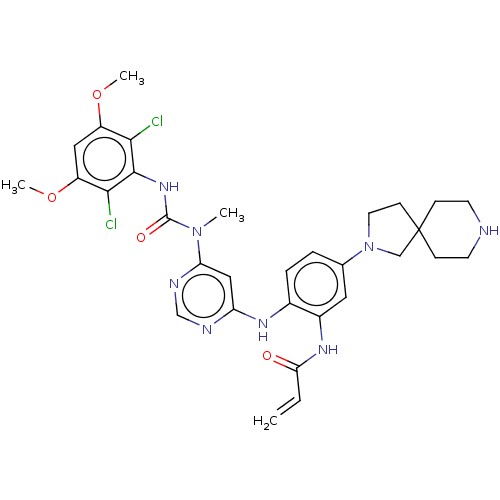
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chen, L; Guan, D; Bai, H; Gou, J; Zhao, W; Wang, Z; Ling, L; Ma, Y Pyrimidine derivative, method for preparing same and use thereof in medicine US Patent US10654836 Publication Date 5/19/2020
Chen, L; Guan, D; Bai, H; Gou, J; Zhao, W; Wang, Z; Ling, L; Ma, Y Pyrimidine derivative, method for preparing same and use thereof in medicine US Patent US10654836 Publication Date 5/19/2020