

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Apelin receptor | ||
| Ligand | BDBM456609 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | GTPγS Binding | ||
| EC50 | 300±n/a nM | ||
| Citation |  Brown, M; Chen, N; Chen, X; Chen, Y; Cheng, AC; Connors, RV; Deignan, J; Dransfield, PJ; Du, X; Fu, Z; Harvey, JS; Heath, JA; Heumann, LV; Houze, J; Kayser, F; Khakoo, AY; Kopecky, D; Lai, S; Ma, Z; Medina, JC; Mihalic, JT; Olson, SH; Pattaropong, V; Swaminath, G; Wang, X; Wanska, M; Yeh, W Triazole furan compounds as agonists of the APJ receptor US Patent US10736883 Publication Date 8/11/2020 Brown, M; Chen, N; Chen, X; Chen, Y; Cheng, AC; Connors, RV; Deignan, J; Dransfield, PJ; Du, X; Fu, Z; Harvey, JS; Heath, JA; Heumann, LV; Houze, J; Kayser, F; Khakoo, AY; Kopecky, D; Lai, S; Ma, Z; Medina, JC; Mihalic, JT; Olson, SH; Pattaropong, V; Swaminath, G; Wang, X; Wanska, M; Yeh, W Triazole furan compounds as agonists of the APJ receptor US Patent US10736883 Publication Date 8/11/2020 | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Apelin receptor | |||
| Name: | Apelin receptor | ||
| Synonyms: | AGTRL1 | APJ | APJ_HUMAN | APLNR | Angiotensin receptor-like 1 | Apelin receptor | Apelin receptor (APJ) | G-protein coupled receptor APJ | G-protein coupled receptor HG11 | ||
| Type: | Enzyme Catalytic Domain | ||
| Mol. Mass.: | 42664.06 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P35414 | ||
| Residue: | 380 | ||
| Sequence: |
| ||
| BDBM456609 | |||
| n/a | |||
| Name | BDBM456609 | ||
| Synonyms: | US10736883, Example 46.0 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C21H22N6O5S | ||
| Mol. Mass. | 470.502 | ||
| SMILES | COc1cccc(OC)c1-n1c(NS(=O)(=O)[C@@H](C)Cc2ccncn2)nnc1-c1ccco1 |r,wU:16.17,(-6.31,-5.56,;-4.82,-5.96,;-3.73,-4.87,;-4.13,-3.38,;-3.04,-2.29,;-1.55,-2.69,;-1.16,-4.18,;.33,-4.58,;1.42,-3.49,;-2.24,-5.27,;-1.61,-6.99,;-.17,-7.55,;1.17,-6.78,;2.5,-7.55,;3.83,-8.32,;1.73,-8.88,;3.27,-6.21,;2.5,-4.88,;4.81,-6.21,;5.58,-4.88,;7.12,-4.88,;7.89,-3.55,;7.12,-2.21,;5.58,-2.21,;4.81,-3.55,;-.25,-9.08,;-1.74,-9.48,;-2.57,-8.19,;-4.06,-7.79,;-4.9,-6.5,;-6.39,-6.9,;-6.47,-8.44,;-5.03,-8.99,)| | ||
| Structure | 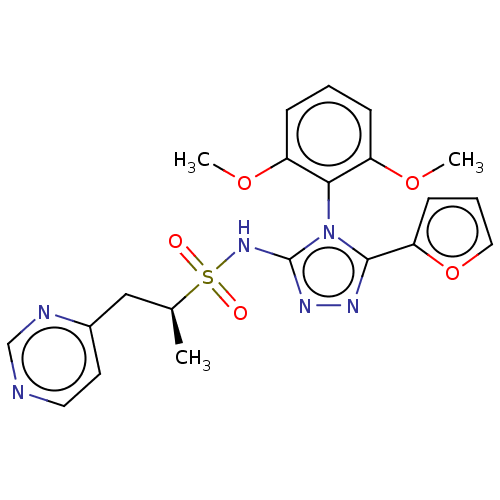
| ||