| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Dihydroorotate dehydrogenase (quinone), mitochondrial |
|---|
| Ligand | BDBM28815 |
|---|
| Substrate/Competitor | BDBM14700 |
|---|
| Meas. Tech. | DHODH Inhibition Assay |
|---|
| pH | 8±n/a |
|---|
| Temperature | 293.15±n/a K |
|---|
| IC50 | 780±250 nM |
|---|
| Citation |  Gujjar, R; Marwaha, A; El Mazouni, F; White, J; White, KL; Creason, S; Shackleford, DM; Baldwin, J; Charman, WN; Buckner, FS; Charman, S; Rathod, PK; Phillips, MA Identification of a metabolically stable triazolopyrimidine-based dihydroorotate dehydrogenase inhibitor with antimalarial activity in mice. J Med Chem52:1864-72 (2009) [PubMed] Article Gujjar, R; Marwaha, A; El Mazouni, F; White, J; White, KL; Creason, S; Shackleford, DM; Baldwin, J; Charman, WN; Buckner, FS; Charman, S; Rathod, PK; Phillips, MA Identification of a metabolically stable triazolopyrimidine-based dihydroorotate dehydrogenase inhibitor with antimalarial activity in mice. J Med Chem52:1864-72 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Dihydroorotate dehydrogenase (quinone), mitochondrial |
|---|
| Name: | Dihydroorotate dehydrogenase (quinone), mitochondrial |
|---|
| Synonyms: | DHOdehase | Dihydroorotate Dehydrogenase (DHODH) | Dihydroorotate dehydrogenase homolog | Dihydroorotate oxidase | PYRD_PLAF7 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65581.88 |
|---|
| Organism: | Plasmodium falciparum (isolate 3D7) |
|---|
| Description: | Q08210 |
|---|
| Residue: | 569 |
|---|
| Sequence: | MISKLKPQFMFLPKKHILSYCRKDVLNLFEQKFYYTSKRKESNNMKNESLLRLINYNRYY
NKIDSNNYYNGGKILSNDRQYIYSPLCEYKKKINDISSYVSVPFKINIRNLGTSNFVNNK
KDVLDNDYIYENIKKEKSKHKKIIFLLFVSLFGLYGFFESYNPEFFLYDIFLKFCLKYID
GEICHDLFLLLGKYNILPYDTSNDSIYACTNIKHLDFINPFGVAAGFDKNGVCIDSILKL
GFSFIEIGTITPRGQTGNAKPRIFRDVESRSIINSCGFNNMGCDKVTENLILFRKRQEED
KLLSKHIVGVSIGKNKDTVNIVDDLKYCINKIGRYADYIAINVSSPNTPGLRDNQEAGKL
KNIILSVKEEIDNLEKNNIMNDESTYNEDNKIVEKKNNFNKNNSHMMKDAKDNFLWFNTT
KKKPLVFVKLAPDLNQEQKKEIADVLLETNIDGMIISNTTTQINDIKSFENKKGGVSGAK
LKDISTKFICEMYNYTNKQIPIIASGGIFSGLDALEKIEAGASVCQLYSCLVFNGMKSAV
QIKRELNHLLYQRGYYNLKEAIGRKHSKS
|
|
|
|---|
| BDBM28815 |
|---|
| BDBM14700 |
|---|
| Name | BDBM28815 |
|---|
| Synonyms: | N-(4-bromophenyl)-5-methyl-[1,2,4]triazolo[1,5-a]pyrimidin-7-amine | triazolopyrimidine-based compound, DSM98 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H10BrN5 |
|---|
| Mol. Mass. | 304.145 |
|---|
| SMILES | Cc1cc(Nc2ccc(Br)cc2)n2ncnc2n1 |
|---|
| Structure | 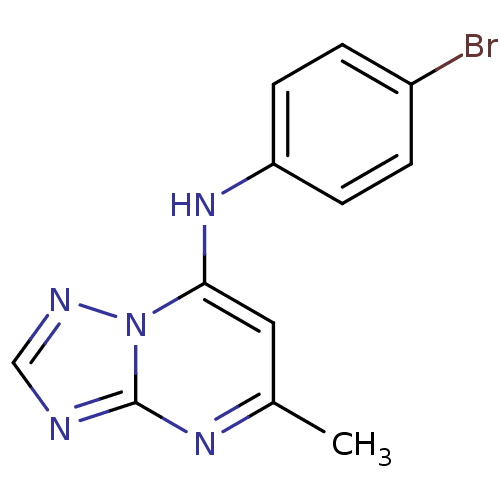
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gujjar, R; Marwaha, A; El Mazouni, F; White, J; White, KL; Creason, S; Shackleford, DM; Baldwin, J; Charman, WN; Buckner, FS; Charman, S; Rathod, PK; Phillips, MA Identification of a metabolically stable triazolopyrimidine-based dihydroorotate dehydrogenase inhibitor with antimalarial activity in mice. J Med Chem52:1864-72 (2009) [PubMed] Article
Gujjar, R; Marwaha, A; El Mazouni, F; White, J; White, KL; Creason, S; Shackleford, DM; Baldwin, J; Charman, WN; Buckner, FS; Charman, S; Rathod, PK; Phillips, MA Identification of a metabolically stable triazolopyrimidine-based dihydroorotate dehydrogenase inhibitor with antimalarial activity in mice. J Med Chem52:1864-72 (2009) [PubMed] Article