| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50459625 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1766900 (CHEMBL4202147) |
|---|
| Ki | >10000±n/a nM |
|---|
| Citation |  Provencher, BA; Eshleman, AJ; Johnson, RA; Shi, X; Kryatova, O; Nelson, J; Tian, J; Gonzalez, M; Meltzer, PC; Janowsky, A Synthesis and Discovery of Arylpiperidinylquinazolines: New Inhibitors of the Vesicular Monoamine Transporter. J Med Chem61:9121-9131 (2018) [PubMed] Article Provencher, BA; Eshleman, AJ; Johnson, RA; Shi, X; Kryatova, O; Nelson, J; Tian, J; Gonzalez, M; Meltzer, PC; Janowsky, A Synthesis and Discovery of Arylpiperidinylquinazolines: New Inhibitors of the Vesicular Monoamine Transporter. J Med Chem61:9121-9131 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM50459625 |
|---|
| n/a |
|---|
| Name | BDBM50459625 |
|---|
| Synonyms: | CHEMBL4205254 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H26FN3O4 |
|---|
| Mol. Mass. | 439.4793 |
|---|
| SMILES | CCOC(=O)C1CN(CCn2c(=O)[nH]c3ccccc3c2=O)CCC1c1ccc(F)cc1 |
|---|
| Structure | 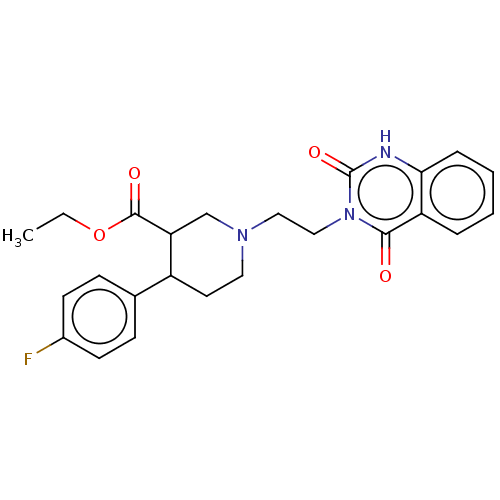
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Provencher, BA; Eshleman, AJ; Johnson, RA; Shi, X; Kryatova, O; Nelson, J; Tian, J; Gonzalez, M; Meltzer, PC; Janowsky, A Synthesis and Discovery of Arylpiperidinylquinazolines: New Inhibitors of the Vesicular Monoamine Transporter. J Med Chem61:9121-9131 (2018) [PubMed] Article
Provencher, BA; Eshleman, AJ; Johnson, RA; Shi, X; Kryatova, O; Nelson, J; Tian, J; Gonzalez, M; Meltzer, PC; Janowsky, A Synthesis and Discovery of Arylpiperidinylquinazolines: New Inhibitors of the Vesicular Monoamine Transporter. J Med Chem61:9121-9131 (2018) [PubMed] Article