| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Amine oxidase [flavin-containing] B |
|---|
| Ligand | BDBM50536257 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1933261 (CHEMBL4478913) |
|---|
| IC50 | 65000±n/a nM |
|---|
| Citation |  Di Pietro, O; Alencar, N; Esteban, G; Viayna, E; Sza?aj, N; V�zquez, J; Ju�rez-Jim�nez, J; Sola, I; P�rez, B; Sol�, M; Unzeta, M; Mu�oz-Torrero, D; Luque, FJ Design, synthesis and biological evaluation of N-methyl-N-[(1,2,3-triazol-4-yl)alkyl]propargylamines as novel monoamine oxidase B inhibitors. Bioorg Med Chem24:4835-4854 (2016) [PubMed] Article Di Pietro, O; Alencar, N; Esteban, G; Viayna, E; Sza?aj, N; V�zquez, J; Ju�rez-Jim�nez, J; Sola, I; P�rez, B; Sol�, M; Unzeta, M; Mu�oz-Torrero, D; Luque, FJ Design, synthesis and biological evaluation of N-methyl-N-[(1,2,3-triazol-4-yl)alkyl]propargylamines as novel monoamine oxidase B inhibitors. Bioorg Med Chem24:4835-4854 (2016) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Amine oxidase [flavin-containing] B |
|---|
| Name: | Amine oxidase [flavin-containing] B |
|---|
| Synonyms: | AOFB_HUMAN | MAO-B | MAOB | Monoamine oxidase type B | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAO B) | Monoamine oxidase type B (MAOB) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 58768.76 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P27338 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MSNKCDVVVVGGGISGMAAAKLLHDSGLNVVVLEARDRVGGRTYTLRNQKVKYVDLGGSY
VGPTQNRILRLAKELGLETYKVNEVERLIHHVKGKSYPFRGPFPPVWNPITYLDHNNFWR
TMDDMGREIPSDAPWKAPLAEEWDNMTMKELLDKLCWTESAKQLATLFVNLCVTAETHEV
SALWFLWYVKQCGGTTRIISTTNGGQERKFVGGSGQVSERIMDLLGDRVKLERPVIYIDQ
TRENVLVETLNHEMYEAKYVISAIPPTLGMKIHFNPPLPMMRNQMITRVPLGSVIKCIVY
YKEPFWRKKDYCGTMIIDGEEAPVAYTLDDTKPEGNYAAIMGFILAHKARKLARLTKEER
LKKLCELYAKVLGSLEALEPVHYEEKNWCEEQYSGGCYTTYFPPGILTQYGRVLRQPVDR
IYFAGTETATHWSGYMEGAVEAGERAAREILHAMGKIPEDEIWQSEPESVDVPAQPITTT
FLERHLPSVPGLLRLIGLTTIFSATALGFLAHKRGLLVRV
|
|
|
|---|
| BDBM50536257 |
|---|
| n/a |
|---|
| Name | BDBM50536257 |
|---|
| Synonyms: | CHEMBL4577856 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H18N4 |
|---|
| Mol. Mass. | 254.3302 |
|---|
| SMILES | CN(CC#C)Cc1cccc(Cc2cn(C)nn2)c1 |
|---|
| Structure | 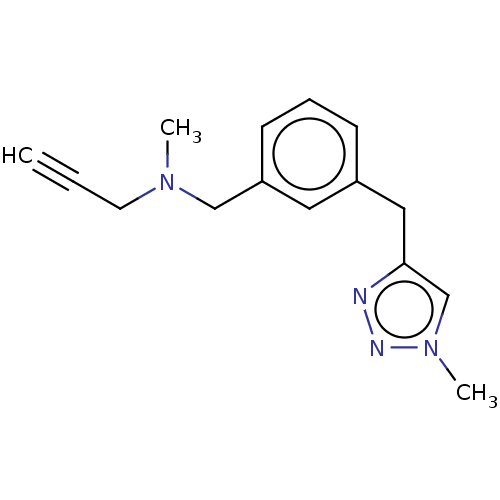
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Di Pietro, O; Alencar, N; Esteban, G; Viayna, E; Sza?aj, N; V�zquez, J; Ju�rez-Jim�nez, J; Sola, I; P�rez, B; Sol�, M; Unzeta, M; Mu�oz-Torrero, D; Luque, FJ Design, synthesis and biological evaluation of N-methyl-N-[(1,2,3-triazol-4-yl)alkyl]propargylamines as novel monoamine oxidase B inhibitors. Bioorg Med Chem24:4835-4854 (2016) [PubMed] Article
Di Pietro, O; Alencar, N; Esteban, G; Viayna, E; Sza?aj, N; V�zquez, J; Ju�rez-Jim�nez, J; Sola, I; P�rez, B; Sol�, M; Unzeta, M; Mu�oz-Torrero, D; Luque, FJ Design, synthesis and biological evaluation of N-methyl-N-[(1,2,3-triazol-4-yl)alkyl]propargylamines as novel monoamine oxidase B inhibitors. Bioorg Med Chem24:4835-4854 (2016) [PubMed] Article