| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor gamma |
|---|
| Ligand | BDBM50615955 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2304837 |
|---|
| EC50 | 240±n/a nM |
|---|
| Citation |  Kato, T; Ohara, T; Suzuki, N; Naya, N; Fukao, K; Tokuyama, R; Muto, S; Fukasawa, H; Itai, A; Matsumura, KI Discovery and structure-activity relationship study of 2-piperazinyl-benzothiazole derivatives as potent and selective PPAR? agonists. Bioorg Med Chem82:0 (2023) [PubMed] Kato, T; Ohara, T; Suzuki, N; Naya, N; Fukao, K; Tokuyama, R; Muto, S; Fukasawa, H; Itai, A; Matsumura, KI Discovery and structure-activity relationship study of 2-piperazinyl-benzothiazole derivatives as potent and selective PPAR? agonists. Bioorg Med Chem82:0 (2023) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor gamma |
|---|
| Name: | Peroxisome proliferator-activated receptor gamma |
|---|
| Synonyms: | Nr1c3 | Nuclear receptor subfamily 1 group C member 3 | PPAR-gamma | PPARG_MOUSE | Pparg |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57593.55 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_1453700 |
|---|
| Residue: | 505 |
|---|
| Sequence: | MGETLGDSPVDPEHGAFADALPMSTSQEITMVDTEMPFWPTNFGISSVDLSVMEDHSHSF
DIKPFTTVDFSSISAPHYEDIPFTRADPMVADYKYDLKLQEYQSAIKVEPASPPYYSEKT
QLYNRPHEEPSNSLMAIECRVCGDKASGFHYGVHACEGCKGFFRRTIRLKLIYDRCDLNC
RIHKKSRNKCQYCRFQKCLAVGMSHNAIRFGRMPQAEKEKLLAEISSDIDQLNPESADLR
ALAKHLYDSYIKSFPLTKAKARAILTGKTTDKSPFVIYDMNSLMMGEDKIKFKHITPLQE
QSKEVAIRIFQGCQFRSVEAVQEITEYAKNIPGFINLDLNDQVTLLKYGVHEIIYTMLAS
LMNKDGVLISEGQGFMTREFLKNLRKPFGDFMEPKFEFAVKFNALELDDSDLAIFIAVII
LSGDRPGLLNVKPIEDIQDNLLQALELQLKLNHPESSQLFAKVLQKMTDLRQIVTEHVQL
LHVIKKTETDMSLHPLLQEIYKDLY
|
|
|
|---|
| BDBM50615955 |
|---|
| n/a |
|---|
| Name | BDBM50615955 |
|---|
| Synonyms: | CHEMBL5283926 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H25ClF3N3O3S |
|---|
| Mol. Mass. | 527.987 |
|---|
| SMILES | CC[C@H]1CN(CCN1CCOc1cc(CC(O)=O)ccc1Cl)c1nc2ccc(cc2s1)C(F)(F)F |r| |
|---|
| Structure | 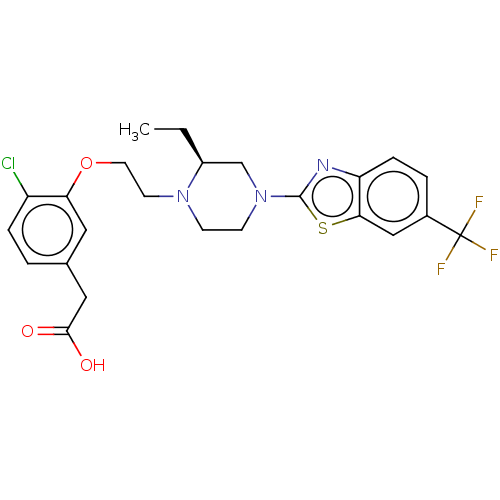
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kato, T; Ohara, T; Suzuki, N; Naya, N; Fukao, K; Tokuyama, R; Muto, S; Fukasawa, H; Itai, A; Matsumura, KI Discovery and structure-activity relationship study of 2-piperazinyl-benzothiazole derivatives as potent and selective PPAR? agonists. Bioorg Med Chem82:0 (2023) [PubMed]
Kato, T; Ohara, T; Suzuki, N; Naya, N; Fukao, K; Tokuyama, R; Muto, S; Fukasawa, H; Itai, A; Matsumura, KI Discovery and structure-activity relationship study of 2-piperazinyl-benzothiazole derivatives as potent and selective PPAR? agonists. Bioorg Med Chem82:0 (2023) [PubMed]