| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cyclin-dependent kinase 2 |
|---|
| Ligand | BDBM50312753 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_615646 (CHEMBL1104220) |
|---|
| IC50 | 607±n/a nM |
|---|
| Citation |  Zeng, Q; Bourbeau, MP; Wohlhieter, GE; Yao, G; Monenschein, H; Rider, JT; Lee, MR; Zhang, S; Lofgren, J; Freeman, D; Li, C; Tominey, E; Huang, X; Hoffman, D; Yamane, H; Tasker, AS; Dominguez, C; Viswanadhan, VN; Hungate, R; Zhang, X 2-Aminothiadiazole inhibitors of AKT1 as potential cancer therapeutics. Bioorg Med Chem Lett20:1652-6 (2010) [PubMed] Article Zeng, Q; Bourbeau, MP; Wohlhieter, GE; Yao, G; Monenschein, H; Rider, JT; Lee, MR; Zhang, S; Lofgren, J; Freeman, D; Li, C; Tominey, E; Huang, X; Hoffman, D; Yamane, H; Tasker, AS; Dominguez, C; Viswanadhan, VN; Hungate, R; Zhang, X 2-Aminothiadiazole inhibitors of AKT1 as potential cancer therapeutics. Bioorg Med Chem Lett20:1652-6 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cyclin-dependent kinase 2 |
|---|
| Name: | Cyclin-dependent kinase 2 |
|---|
| Synonyms: | CDK2 | CDK2-Kinase | CDK2_HUMAN | CDKN2 | Cell division protein kinase 2 | Cyclin-dependent kinase 2 (CDK2) | Protein cereblon/Cyclin-dependent kinase 2 | p33 protein kinase |
|---|
| Type: | Enzyme Subunit |
|---|
| Mol. Mass.: | 33938.17 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P24941 |
|---|
| Residue: | 298 |
|---|
| Sequence: | MENFQKVEKIGEGTYGVVYKARNKLTGEVVALKKIRLDTETEGVPSTAIREISLLKELNH
PNIVKLLDVIHTENKLYLVFEFLHQDLKKFMDASALTGIPLPLIKSYLFQLLQGLAFCHS
HRVLHRDLKPQNLLINTEGAIKLADFGLARAFGVPVRTYTHEVVTLWYRAPEILLGCKYY
STAVDIWSLGCIFAEMVTRRALFPGDSEIDQLFRIFRTLGTPDEVVWPGVTSMPDYKPSF
PKWARQDFSKVVPPLDEDGRSLLSQMLHYDPNKRISAKAALAHPFFQDVTKPVPHLRL
|
|
|
|---|
| BDBM50312753 |
|---|
| n/a |
|---|
| Name | BDBM50312753 |
|---|
| Synonyms: | (S)-N1-(5-(3-methylisoquinolin-6-yl)-1,3,4-thiadiazol-2-yl)-3-(4-(trifluoromethyl)phenyl)propane-1,2-diamine | CHEMBL1081629 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H20F3N5S |
|---|
| Mol. Mass. | 443.488 |
|---|
| SMILES | Cc1cc2cc(ccc2cn1)-c1nnc(NC[C@@H](N)Cc2ccc(cc2)C(F)(F)F)s1 |r| |
|---|
| Structure | 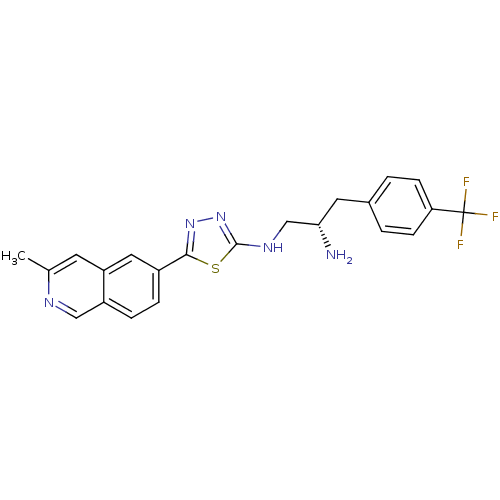
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zeng, Q; Bourbeau, MP; Wohlhieter, GE; Yao, G; Monenschein, H; Rider, JT; Lee, MR; Zhang, S; Lofgren, J; Freeman, D; Li, C; Tominey, E; Huang, X; Hoffman, D; Yamane, H; Tasker, AS; Dominguez, C; Viswanadhan, VN; Hungate, R; Zhang, X 2-Aminothiadiazole inhibitors of AKT1 as potential cancer therapeutics. Bioorg Med Chem Lett20:1652-6 (2010) [PubMed] Article
Zeng, Q; Bourbeau, MP; Wohlhieter, GE; Yao, G; Monenschein, H; Rider, JT; Lee, MR; Zhang, S; Lofgren, J; Freeman, D; Li, C; Tominey, E; Huang, X; Hoffman, D; Yamane, H; Tasker, AS; Dominguez, C; Viswanadhan, VN; Hungate, R; Zhang, X 2-Aminothiadiazole inhibitors of AKT1 as potential cancer therapeutics. Bioorg Med Chem Lett20:1652-6 (2010) [PubMed] Article