| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Lysophosphatidic acid receptor 1 |
|---|
| Ligand | BDBM50233166 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1649796 (CHEMBL3998930) |
|---|
| IC50 | 18000±n/a nM |
|---|
| Citation |  Banerjee, S; Norman, DD; Lee, SC; Parrill, AL; Pham, TC; Baker, DL; Tigyi, GJ; Miller, DD Highly Potent Non-Carboxylic Acid Autotaxin Inhibitors Reduce Melanoma Metastasis and Chemotherapeutic Resistance of Breast Cancer Stem Cells. J Med Chem60:1309-1324 (2017) [PubMed] Article Banerjee, S; Norman, DD; Lee, SC; Parrill, AL; Pham, TC; Baker, DL; Tigyi, GJ; Miller, DD Highly Potent Non-Carboxylic Acid Autotaxin Inhibitors Reduce Melanoma Metastasis and Chemotherapeutic Resistance of Breast Cancer Stem Cells. J Med Chem60:1309-1324 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Lysophosphatidic acid receptor 1 |
|---|
| Name: | Lysophosphatidic acid receptor 1 |
|---|
| Synonyms: | EDG2 | LPA receptor 1 | LPA-1 | LPA1 | LPAR1 | LPAR1_HUMAN | Lysophosphatidic acid receptor 1 (LPA1) | Lysophosphatidic acid receptor Edg-2 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 41120.55 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q92633 |
|---|
| Residue: | 364 |
|---|
| Sequence: | MAAISTSIPVISQPQFTAMNEPQCFYNESIAFFYNRSGKHLATEWNTVSKLVMGLGITVC
IFIMLANLLVMVAIYVNRRFHFPIYYLMANLAAADFFAGLAYFYLMFNTGPNTRRLTVST
WLLRQGLIDTSLTASVANLLAIAIERHITVFRMQLHTRMSNRRVVVVIVVIWTMAIVMGA
IPSVGWNCICDIENCSNMAPLYSDSYLVFWAIFNLVTFVVMVVLYAHIFGYVRQRTMRMS
RHSSGPRRNRDTMMSLLKTVVIVLGAFIICWTPGLVLLLLDVCCPQCDVLAYEKFFLLLA
EFNSAMNPIIYSYRDKEMSATFRQILCCQRSENPTGPTEGSDRSASSLNHTILAGVHSND
HSVV
|
|
|
|---|
| BDBM50233166 |
|---|
| n/a |
|---|
| Name | BDBM50233166 |
|---|
| Synonyms: | CHEMBL4060128 | US11124490, Compound 3g |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H13Cl4FN2O4S |
|---|
| Mol. Mass. | 502.172 |
|---|
| SMILES | Fc1c(Cl)cc(NC(=O)c2cc(c(Cl)cc2Cl)S(=O)(=O)N2CCOCC2)cc1Cl |
|---|
| Structure | 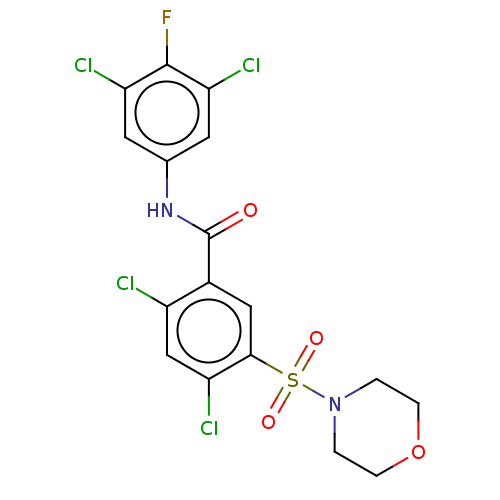
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Banerjee, S; Norman, DD; Lee, SC; Parrill, AL; Pham, TC; Baker, DL; Tigyi, GJ; Miller, DD Highly Potent Non-Carboxylic Acid Autotaxin Inhibitors Reduce Melanoma Metastasis and Chemotherapeutic Resistance of Breast Cancer Stem Cells. J Med Chem60:1309-1324 (2017) [PubMed] Article
Banerjee, S; Norman, DD; Lee, SC; Parrill, AL; Pham, TC; Baker, DL; Tigyi, GJ; Miller, DD Highly Potent Non-Carboxylic Acid Autotaxin Inhibitors Reduce Melanoma Metastasis and Chemotherapeutic Resistance of Breast Cancer Stem Cells. J Med Chem60:1309-1324 (2017) [PubMed] Article