| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Signal transducer and activator of transcription 3 |
|---|
| Ligand | BDBM50189922 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Bioassay Evaluation |
|---|
| IC50 | 6800±3700 nM |
|---|
| Citation |  Huryn, DM; Wipf, P; Grandis, JR; LaPorte, MG; Johnston, PA; Schurdak, ME; Colombo, R 6-aryl-7-substituted-3-(1H-pyrazol-5-yl)-7H-[1,2,4]triazolo[3,4-B][1,3,4]thiadiazines as inhibitors of the STAT3 pathway with anti-proliferative activity US Patent US10618914 Publication Date 4/14/2020 Huryn, DM; Wipf, P; Grandis, JR; LaPorte, MG; Johnston, PA; Schurdak, ME; Colombo, R 6-aryl-7-substituted-3-(1H-pyrazol-5-yl)-7H-[1,2,4]triazolo[3,4-B][1,3,4]thiadiazines as inhibitors of the STAT3 pathway with anti-proliferative activity US Patent US10618914 Publication Date 4/14/2020 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Signal transducer and activator of transcription 3 |
|---|
| Name: | Signal transducer and activator of transcription 3 |
|---|
| Synonyms: | APRF | STAT3 | STAT3_HUMAN | Signal transducer and activator of transcription 3 | Signal transducer and activator of transcription 3 (STAT3) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 88064.31 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P40763 |
|---|
| Residue: | 770 |
|---|
| Sequence: | MAQWNQLQQLDTRYLEQLHQLYSDSFPMELRQFLAPWIESQDWAYAASKESHATLVFHNL
LGEIDQQYSRFLQESNVLYQHNLRRIKQFLQSRYLEKPMEIARIVARCLWEESRLLQTAA
TAAQQGGQANHPTAAVVTEKQQMLEQHLQDVRKRVQDLEQKMKVVENLQDDFDFNYKTLK
SQGDMQDLNGNNQSVTRQKMQQLEQMLTALDQMRRSIVSELAGLLSAMEYVQKTLTDEEL
ADWKRRQQIACIGGPPNICLDRLENWITSLAESQLQTRQQIKKLEELQQKVSYKGDPIVQ
HRPMLEERIVELFRNLMKSAFVVERQPCMPMHPDRPLVIKTGVQFTTKVRLLVKFPELNY
QLKIKVCIDKDSGDVAALRGSRKFNILGTNTKVMNMEESNNGSLSAEFKHLTLREQRCGN
GGRANCDASLIVTEELHLITFETEVYHQGLKIDLETHSLPVVVISNICQMPNAWASILWY
NMLTNNPKNVNFFTKPPIGTWDQVAEVLSWQFSSTTKRGLSIEQLTTLAEKLLGPGVNYS
GCQITWAKFCKENMAGKGFSFWVWLDNIIDLVKKYILALWNEGYIMGFISKERERAILST
KPPGTFLLRFSESSKEGGVTFTWVEKDISGKTQIQSVEPYTKQQLNNMSFAEIIMGYKIM
DATNILVSPLVYLYPDIPKEEAFGKYCRPESQEHPEADPGSAAPYLKTKFICVTPTTCSN
TIDLPMSPRTLDSLMQFGNNGEGAEPSAGGQFESLTFDMELTSECATSPM
|
|
|
|---|
| BDBM50189922 |
|---|
| n/a |
|---|
| Name | BDBM50189922 |
|---|
| Synonyms: | CHEMBL1527817 | US10618914, Compound 1a | US11111253, Compound 1a |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H12Cl2N6S |
|---|
| Mol. Mass. | 391.278 |
|---|
| SMILES | Clc1ccc(cc1Cl)C1=Nn2c(SC1)nnc2-c1[nH]nc2CCCc12 |t:9| |
|---|
| Structure | 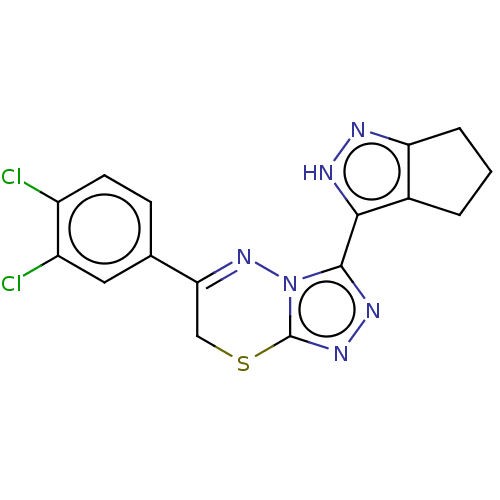
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Huryn, DM; Wipf, P; Grandis, JR; LaPorte, MG; Johnston, PA; Schurdak, ME; Colombo, R 6-aryl-7-substituted-3-(1H-pyrazol-5-yl)-7H-[1,2,4]triazolo[3,4-B][1,3,4]thiadiazines as inhibitors of the STAT3 pathway with anti-proliferative activity US Patent US10618914 Publication Date 4/14/2020
Huryn, DM; Wipf, P; Grandis, JR; LaPorte, MG; Johnston, PA; Schurdak, ME; Colombo, R 6-aryl-7-substituted-3-(1H-pyrazol-5-yl)-7H-[1,2,4]triazolo[3,4-B][1,3,4]thiadiazines as inhibitors of the STAT3 pathway with anti-proliferative activity US Patent US10618914 Publication Date 4/14/2020