| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 4F2 |
|---|
| Ligand | BDBM558218 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Inhibition Test for Each Compound of the Present Invention Against 20-HETE Producing Enzymes (CYP4F2 and CYP4A11) |
|---|
| IC50 | 6.20±n/a nM |
|---|
| Citation |  Tanaka, H; Kawamura, M; Hamada, M; Kobashi, Y; Ito, Y; Suzuki, K; Bohno, A; Funayama, K Pyridine compound substituted with azole US Patent US11365192 Publication Date 6/21/2022 Tanaka, H; Kawamura, M; Hamada, M; Kobashi, Y; Ito, Y; Suzuki, K; Bohno, A; Funayama, K Pyridine compound substituted with azole US Patent US11365192 Publication Date 6/21/2022 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 4F2 |
|---|
| Name: | Cytochrome P450 4F2 |
|---|
| Synonyms: | 20-HETE synthase | 20-hydroxyeicosatetraenoic acid synthase | Arachidonic acid omega-hydroxylase | CP4F2_HUMAN | CYP4F2 | CYPIVF2 | Cytochrome P450 4F2 | Cytochrome P450-LTB-omega | Leukotriene-B(4) 20-monooxygenase 1 | Leukotriene-B(4) omega-hydroxylase 1 | Phylloquinone omega-hydroxylase CYP4F2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 59859.32 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_10743 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MSQLSLSWLGLWPVAASPWLLLLLVGASWLLAHVLAWTYAFYDNCRRLRCFPQPPRRNWF
WGHQGMVNPTEEGMRVLTQLVATYPQGFKVWMGPISPLLSLCHPDIIRSVINASAAIAPK
DKFFYSFLEPWLGDGLLLSAGDKWSRHRRMLTPAFHFNILKPYMKIFNESVNIMHAKWQL
LASEGSACLDMFEHISLMTLDSLQKCVFSFDSHCQEKPSEYIAAILELSALVSKRHHEIL
LHIDFLYYLTPDGQRFRRACRLVHDFTDAVIQERRRTLPSQGVDDFLQAKAKSKTLDFID
VLLLSKDEDGKKLSDEDIRAEADTFMFEGHDTTASGLSWVLYHLAKHPEYQERCRQEVQE
LLKDREPKEIEWDDLAHLPFLTMCMKESLRLHPPVPVISRHVTQDIVLPDGRVIPKGIIC
LISVFGTHHNPAVWPDPEVYDPFRFDPENIKERSPLAFIPFSAGPRNCIGQTFAMAEMKV
VLALTLLRFRVLPDHTEPRRKPELVLRAEGGLWLRVEPLS
|
|
|
|---|
| BDBM558218 |
|---|
| n/a |
|---|
| Name | BDBM558218 |
|---|
| Synonyms: | 2-Methyl-5-[[6-(1H-pyrazol-5-yl)-3-pyridinyl]oxymethyl]phenol | US11365192, Example 12-1 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H15N3O2 |
|---|
| Mol. Mass. | 281.3092 |
|---|
| SMILES | Cc1ccc(COc2ccc(nc2)-c2ccn[nH]2)cc1O |
|---|
| Structure | 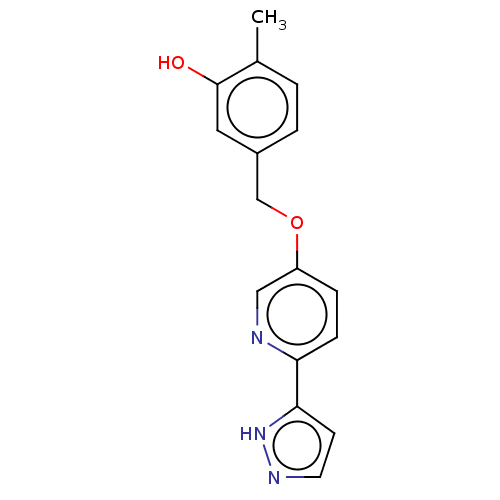
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tanaka, H; Kawamura, M; Hamada, M; Kobashi, Y; Ito, Y; Suzuki, K; Bohno, A; Funayama, K Pyridine compound substituted with azole US Patent US11365192 Publication Date 6/21/2022
Tanaka, H; Kawamura, M; Hamada, M; Kobashi, Y; Ito, Y; Suzuki, K; Bohno, A; Funayama, K Pyridine compound substituted with azole US Patent US11365192 Publication Date 6/21/2022