| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM319614 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | 5-HT2A Receptor Antagonism Activity Test |
|---|
| IC50 | 6.36±n/a nM |
|---|
| Citation |  Jiang, H; Wang, Z; Li, J; Zhang, R; He, Y; Liu, Y; Bi, M; Liu, Z; Tian, G; Chen, W; Yang, F; Wu, C; Wang, Y; Jiang, X; Yin, J; Wang, G; Shen, J Heterocyclic compounds, process for preparation of the same and use thereof US Patent US10174011 Publication Date 1/8/2019 Jiang, H; Wang, Z; Li, J; Zhang, R; He, Y; Liu, Y; Bi, M; Liu, Z; Tian, G; Chen, W; Yang, F; Wu, C; Wang, Y; Jiang, X; Yin, J; Wang, G; Shen, J Heterocyclic compounds, process for preparation of the same and use thereof US Patent US10174011 Publication Date 1/8/2019 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A |
|---|
| Type: | undefined |
|---|
| Mol. Mass.: | 52607.65 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28223 |
|---|
| Residue: | 471 |
|---|
| Sequence: | MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGC
LSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYK
SSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
|
|
|
|---|
| BDBM319614 |
|---|
| n/a |
|---|
| Name | BDBM319614 |
|---|
| Synonyms: | 5-(2-(4-(benzo[b]thiophen-4-yl)piperazin-1-yl)ethyl)indolin-2-one | US10174011, Example 8 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H23N3OS |
|---|
| Mol. Mass. | 377.503 |
|---|
| SMILES | O=C1Cc2cc(CCN3CCN(CC3)c3cccc4sccc34)ccc2N1 |
|---|
| Structure | 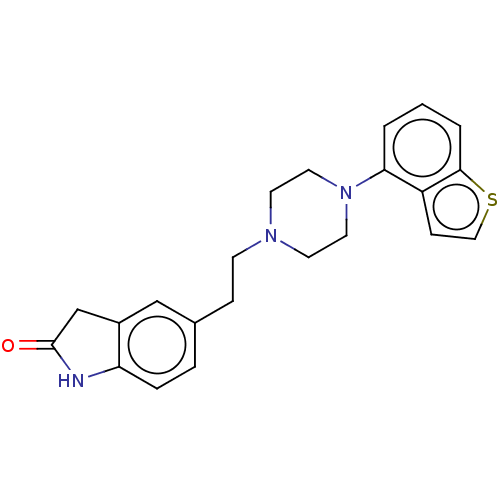
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jiang, H; Wang, Z; Li, J; Zhang, R; He, Y; Liu, Y; Bi, M; Liu, Z; Tian, G; Chen, W; Yang, F; Wu, C; Wang, Y; Jiang, X; Yin, J; Wang, G; Shen, J Heterocyclic compounds, process for preparation of the same and use thereof US Patent US10174011 Publication Date 1/8/2019
Jiang, H; Wang, Z; Li, J; Zhang, R; He, Y; Liu, Y; Bi, M; Liu, Z; Tian, G; Chen, W; Yang, F; Wu, C; Wang, Y; Jiang, X; Yin, J; Wang, G; Shen, J Heterocyclic compounds, process for preparation of the same and use thereof US Patent US10174011 Publication Date 1/8/2019