| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase EZH2 |
|---|
| Ligand | BDBM335713 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | Histone Methyl Transferase Assay |
|---|
| EC50 | <500±n/a nM |
|---|
| Citation |  Kim, KS; Zhang, L; Purandare, AV; Seitz, SP Inhibitors of lysine methyl transferase US Patent US9738630 Publication Date 8/22/2017 Kim, KS; Zhang, L; Purandare, AV; Seitz, SP Inhibitors of lysine methyl transferase US Patent US9738630 Publication Date 8/22/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase EZH2 |
|---|
| Name: | Histone-lysine N-methyltransferase EZH2 |
|---|
| Synonyms: | ENX-1 | EZH2 | EZH2_HUMAN | Enhancer of zeste homolog 2 (EZH2) | Histone-lysine N-methyltransferase EZH2 | KMT6 | Lysine N-methyltransferase 6 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 85367.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15910 |
|---|
| Residue: | 746 |
|---|
| Sequence: | MGQTGKKSEKGPVCWRKRVKSEYMRLRQLKRFRRADEVKSMFSSNRQKILERTEILNQEW
KQRRIQPVHILTSVSSLRGTRECSVTSDLDFPTQVIPLKTLNAVASVPIMYSWSPLQQNF
MVEDETVLHNIPYMGDEVLDQDGTFIEELIKNYDGKVHGDRECGFINDEIFVELVNALGQ
YNDDDDDDDGDDPEEREEKQKDLEDHRDDKESRPPRKFPSDKIFEAISSMFPDKGTAEEL
KEKYKELTEQQLPGALPPECTPNIDGPNAKSVQREQSLHSFHTLFCRRCFKYDCFLHPFH
ATPNTYKRKNTETALDNKPCGPQCYQHLEGAKEFAAALTAERIKTPPKRPGGRRRGRLPN
NSSRPSTPTINVLESKDTDSDREAGTETGGENNDKEEEEKKDETSSSSEANSRCQTPIKM
KPNIEPPENVEWSGAEASMFRVLIGTYYDNFCAIARLIGTKTCRQVYEFRVKESSIIAPA
PAEDVDTPPRKKKRKHRLWAAHCRKIQLKKDGSSNHVYNYQPCDHPRQPCDSSCPCVIAQ
NFCEKFCQCSSECQNRFPGCRCKAQCNTKQCPCYLAVRECDPDLCLTCGAADHWDSKNVS
CKNCSIQRGSKKHLLLAPSDVAGWGIFIKDPVQKNEFISEYCGEIISQDEADRRGKVYDK
YMCSFLFNLNNDFVVDATRKGNKIRFANHSVNPNCYAKVMMVNGDHRIGIFAKRAIQTGE
ELFFDYRYSQADALKYVGIEREMEIP
|
|
|
|---|
| BDBM335713 |
|---|
| n/a |
|---|
| Name | BDBM335713 |
|---|
| Synonyms: | 4'-((1,1-Dioxidothiomorpholino) methyl)-5-(ethyl(tetrahydro-2H- pyran-4-yl)amino)-4-methyl-N- ((1-methyl-3-oxo-2,3,5,6,7,8- hexahydroisoquinolin-4- yl)methyl)-[1,1'-biphenyl]-3- carboxamide | US9738630, Example 8 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H48N4O5S |
|---|
| Mol. Mass. | 660.866 |
|---|
| SMILES | CCN(C1CCOCC1)c1cc(cc(C(=O)NCc2c3CCCCc3c(C)[nH]c2=O)c1C)-c1ccc(CN2CCS(=O)(=O)CC2)cc1 |
|---|
| Structure | 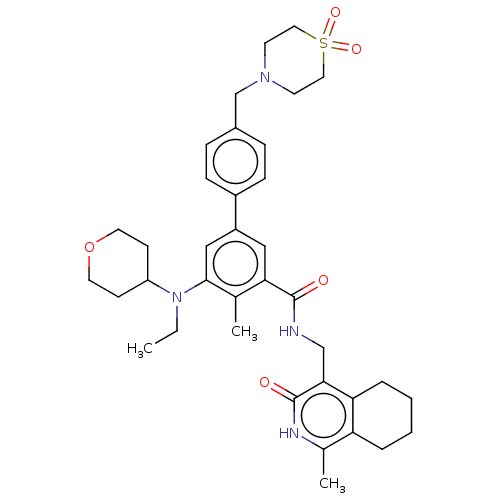
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kim, KS; Zhang, L; Purandare, AV; Seitz, SP Inhibitors of lysine methyl transferase US Patent US9738630 Publication Date 8/22/2017
Kim, KS; Zhang, L; Purandare, AV; Seitz, SP Inhibitors of lysine methyl transferase US Patent US9738630 Publication Date 8/22/2017