| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Aldo-keto reductase family 1 member A1 |
|---|
| Ligand | BDBM16314 |
|---|
| Substrate/Competitor | BDBM16241 |
|---|
| Meas. Tech. | Enzyme Inhibition Assay |
|---|
| IC50 | 1940±n/a nM |
|---|
| Citation |  Van Zandt, MC; Jones, ML; Gunn, DE; Geraci, LS; Jones, JH; Sawicki, DR; Sredy, J; Jacot, JL; Dicioccio, AT; Petrova, T; Mitschler, A; Podjarny, AD Discovery of 3-[(4,5,7-trifluorobenzothiazol-2-yl)methyl]indole-N-acetic acid (lidorestat) and congeners as highly potent and selective inhibitors of aldose reductase for treatment of chronic diabetic complications. J Med Chem48:3141-52 (2005) [PubMed] Article Van Zandt, MC; Jones, ML; Gunn, DE; Geraci, LS; Jones, JH; Sawicki, DR; Sredy, J; Jacot, JL; Dicioccio, AT; Petrova, T; Mitschler, A; Podjarny, AD Discovery of 3-[(4,5,7-trifluorobenzothiazol-2-yl)methyl]indole-N-acetic acid (lidorestat) and congeners as highly potent and selective inhibitors of aldose reductase for treatment of chronic diabetic complications. J Med Chem48:3141-52 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Aldo-keto reductase family 1 member A1 |
|---|
| Name: | Aldo-keto reductase family 1 member A1 |
|---|
| Synonyms: | AK1A1_HUMAN | AKR1A1 | ALDR1 | ALR | Alcohol dehydrogenase [NADP+] | Aldehyde Reductase (ALR1) | Aldehyde reductase | Aldo-keto reductase family 1 member A1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 36574.11 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 325 |
|---|
| Sequence: | MAASCVLLHTGQKMPLIGLGTWKSEPGQVKAAVKYALSVGYRHIDCAAIYGNEPEIGEAL
KEDVGPGKAVPREELFVTSKLWNTKHHPEDVEPALRKTLADLQLEYLDLYLMHWPYAFER
GDNPFPKNADGTICYDSTHYKETWKALEALVAKGLVQALGLSNFNSRQIDDILSVASVRP
AVLQVECHPYLAQNELIAHCQARGLEVTAYSPLGSSDRAWRDPDEPVLLEEPVVLALAEK
YGRSPAQILLRWQVQRKVICIPKSITPSRILQNIKVFDFTFSPEEMKQLNALNKNWRYIV
PMLTVDGKRVPRDAGHPLYPFNDPY
|
|
|
|---|
| BDBM16314 |
|---|
| BDBM16241 |
|---|
| Name | BDBM16314 |
|---|
| Synonyms: | 2-{[6-methoxy-5-(trifluoromethyl)naphthalen-1-yl]-N-methylmethanethioamido}acetic acid | Alredase | CHEMBL436 | N-{[6-methoxy-5-(trifluoromethyl)naphthalen-1-yl]carbonothioyl}-N-methylglycine | Tolrestat |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H14F3NO3S |
|---|
| Mol. Mass. | 357.347 |
|---|
| SMILES | COc1ccc2c(cccc2c1C(F)(F)F)C(=S)N(C)CC(O)=O |
|---|
| Structure | 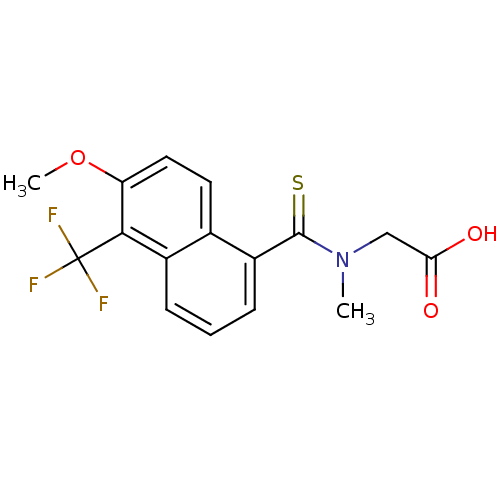
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Van Zandt, MC; Jones, ML; Gunn, DE; Geraci, LS; Jones, JH; Sawicki, DR; Sredy, J; Jacot, JL; Dicioccio, AT; Petrova, T; Mitschler, A; Podjarny, AD Discovery of 3-[(4,5,7-trifluorobenzothiazol-2-yl)methyl]indole-N-acetic acid (lidorestat) and congeners as highly potent and selective inhibitors of aldose reductase for treatment of chronic diabetic complications. J Med Chem48:3141-52 (2005) [PubMed] Article
Van Zandt, MC; Jones, ML; Gunn, DE; Geraci, LS; Jones, JH; Sawicki, DR; Sredy, J; Jacot, JL; Dicioccio, AT; Petrova, T; Mitschler, A; Podjarny, AD Discovery of 3-[(4,5,7-trifluorobenzothiazol-2-yl)methyl]indole-N-acetic acid (lidorestat) and congeners as highly potent and selective inhibitors of aldose reductase for treatment of chronic diabetic complications. J Med Chem48:3141-52 (2005) [PubMed] Article