| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Leukotriene A-4 hydrolase |
|---|
| Ligand | BDBM24262 |
|---|
| Substrate/Competitor | BDBM23991 |
|---|
| Meas. Tech. | Peptidase Assay |
|---|
| pH | 7.5±n/a |
|---|
| Temperature | 295.15±n/a K |
|---|
| IC50 | 25±n/a nM |
|---|
| Km | 1000000±n/a nM |
|---|
| Citation |  Kirkland, TA; Adler, M; Bauman, JG; Chen, M; Haeggström, JZ; King, B; Kochanny, MJ; Liang, AM; Mendoza, L; Phillips, GB; Thunnissen, M; Trinh, L; Whitlow, M; Ye, B; Ye, H; Parkinson, J; Guilford, WJ Synthesis of glutamic acid analogs as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem16:4963-83 (2008) [PubMed] Article Kirkland, TA; Adler, M; Bauman, JG; Chen, M; Haeggström, JZ; King, B; Kochanny, MJ; Liang, AM; Mendoza, L; Phillips, GB; Thunnissen, M; Trinh, L; Whitlow, M; Ye, B; Ye, H; Parkinson, J; Guilford, WJ Synthesis of glutamic acid analogs as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem16:4963-83 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Leukotriene A-4 hydrolase |
|---|
| Name: | Leukotriene A-4 hydrolase |
|---|
| Synonyms: | LKHA4_HUMAN | LTA-4 hydrolase | LTA4 | LTA4H | Leukotriene A(4) hydrolase | Leukotriene A-4 hydrolase (LTA4H) | Leukotriene A4 hydrolase |
|---|
| Type: | Hydrolase; metalloprotease |
|---|
| Mol. Mass.: | 69280.41 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Human recombinant LTA4H. |
|---|
| Residue: | 611 |
|---|
| Sequence: | MPEIVDTCSLASPASVCRTKHLHLRCSVDFTRRTLTGTAALTVQSQEDNLRSLVLDTKDL
TIEKVVINGQEVKYALGERQSYKGSPMEISLPIALSKNQEIVIEISFETSPKSSALQWLT
PEQTSGKEHPYLFSQCQAIHCRAILPCQDTPSVKLTYTAEVSVPKELVALMSAIRDGETP
DPEDPSRKIYKFIQKVPIPCYLIALVVGALESRQIGPRTLVWSEKEQVEKSAYEFSETES
MLKIAEDLGGPYVWGQYDLLVLPPSFPYGGMENPCLTFVTPTLLAGDKSLSNVIAHEISH
SWTGNLVTNKTWDHFWLNEGHTVYLERHICGRLFGEKFRHFNALGGWGELQNSVKTFGET
HPFTKLVVDLTDIDPDVAYSSVPYEKGFALLFYLEQLLGGPEIFLGFLKAYVEKFSYKSI
TTDDWKDFLYSYFKDKVDVLNQVDWNAWLYSPGLPPIKPNYDMTLTNACIALSQRWITAK
EDDLNSFNATDLKDLSSHQLNEFLAQTLQRAPLPLGHIKRMQEVYNFNAINNSEIRFRWL
RLCIQSKWEDAIPLALKMATEQGRMKFTRPLFKDLAAFDKSHDQAVRTYQEHKASMHPVT
AMLVGKDLKVD
|
|
|
|---|
| BDBM24262 |
|---|
| BDBM23991 |
|---|
| Name | BDBM24262 |
|---|
| Synonyms: | (2S)-2-amino-N-[4-(benzyloxy)phenyl]-N'-hydroxypentanediamide | Modified amino acid analog, 6d |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H21N3O4 |
|---|
| Mol. Mass. | 343.377 |
|---|
| SMILES | N[C@@H](CCC(=O)Nc1ccc(OCc2ccccc2)cc1)C(=O)NO |r| |
|---|
| Structure | 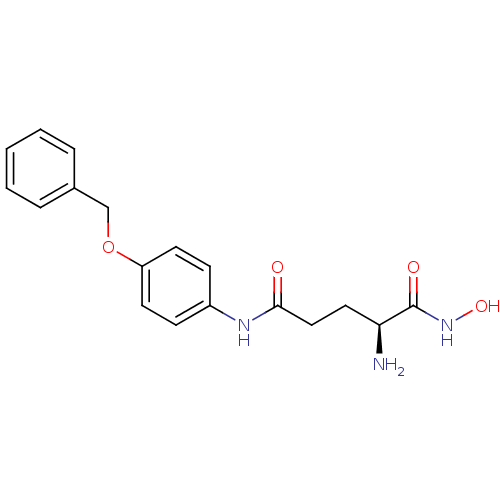
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kirkland, TA; Adler, M; Bauman, JG; Chen, M; Haeggström, JZ; King, B; Kochanny, MJ; Liang, AM; Mendoza, L; Phillips, GB; Thunnissen, M; Trinh, L; Whitlow, M; Ye, B; Ye, H; Parkinson, J; Guilford, WJ Synthesis of glutamic acid analogs as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem16:4963-83 (2008) [PubMed] Article
Kirkland, TA; Adler, M; Bauman, JG; Chen, M; Haeggström, JZ; King, B; Kochanny, MJ; Liang, AM; Mendoza, L; Phillips, GB; Thunnissen, M; Trinh, L; Whitlow, M; Ye, B; Ye, H; Parkinson, J; Guilford, WJ Synthesis of glutamic acid analogs as potent inhibitors of leukotriene A4 hydrolase. Bioorg Med Chem16:4963-83 (2008) [PubMed] Article