| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Fatty acid synthase [2202-2509] |
|---|
| Ligand | BDBM24580 |
|---|
| Substrate/Competitor | BDBM24568 |
|---|
| Meas. Tech. | Fluorogenic Assay for Detection of FASTE Inhibition |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 310.15±n/a K |
|---|
| IC50 | 500±120 nM |
|---|
| Citation |  Richardson, RD; Ma, G; Oyola, Y; Zancanella, M; Knowles, LM; Cieplak, P; Romo, D; Smith, JW Synthesis of novel beta-lactone inhibitors of fatty acid synthase. J Med Chem51:5285-96 (2008) [PubMed] Article Richardson, RD; Ma, G; Oyola, Y; Zancanella, M; Knowles, LM; Cieplak, P; Romo, D; Smith, JW Synthesis of novel beta-lactone inhibitors of fatty acid synthase. J Med Chem51:5285-96 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Inhibition_Run data, Solution Info, Assay Method |
|---|
| |
| Fatty acid synthase [2202-2509] |
|---|
| Name: | Fatty acid synthase [2202-2509] |
|---|
| Synonyms: | FAS | FASN | FAS_HUMAN | Fatty Acid Synthase |
|---|
| Type: | Thioesterase domain |
|---|
| Mol. Mass.: | 33927.11 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | The recombinant thioesterase domain (residues 2202-2509) of FAS was cloned and expressed in Escheria coli. The thioesterase was purified by Ni-affinity chromatography, and analyzed for activity and inhibition by Orlistat. |
|---|
| Residue: | 308 |
|---|
| Sequence: | CPTPKEDGLAQQQTQLNLRSLLVNPEGPTLMRLNSVQSSERPLFLVHPIEGSTTVFHSLA
SRLSIPTYGLQCTRAAPLDSIHSLAAYYIDCIRQVQPEGPYRVAGYSYGACVAFEMCSQL
QAQQSPAPTHNSLFLFDGSPTYVLAYTQSYRAKLTPGCEAEAETEAICFFVQQFTDMEHN
RVLEALLPLKGLEERVAAAVDLIIKSHQGLDRQELSFAARSFYYKLRAAEQYTPKAKYHG
NVMLLRAKTGGAYGEDLGADYNLSQVCDGKVSVHVIEGDHRTLLEGSGLESIISIIHSSL
AEPRVSVR
|
|
|
|---|
| BDBM24580 |
|---|
| BDBM24568 |
|---|
| Name | BDBM24580 |
|---|
| Synonyms: | (2S,5E)-6-(4-fluorophenyl)-1-[(2S,3S)-3-hexyl-4-oxooxetan-2-yl]hex-5-en-2-yl (2S)-2-formamido-4-methylpentanoate | Orlistat derivative, 21c |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H40FNO5 |
|---|
| Mol. Mass. | 489.6193 |
|---|
| SMILES | CCCCCC[C@H]1[C@H](C[C@H](CC\C=C\c2ccc(F)cc2)OC(=O)[C@H](CC(C)C)NC=O)OC1=O |r| |
|---|
| Structure | 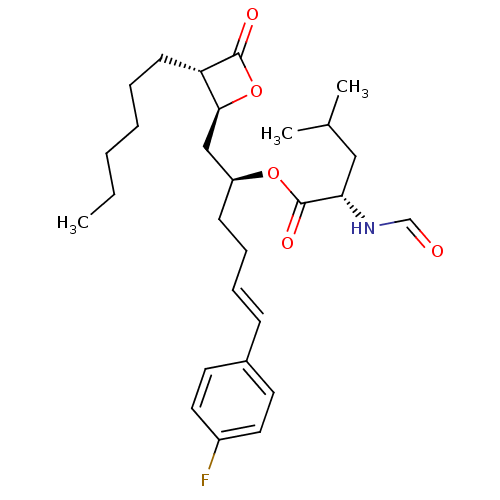
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Richardson, RD; Ma, G; Oyola, Y; Zancanella, M; Knowles, LM; Cieplak, P; Romo, D; Smith, JW Synthesis of novel beta-lactone inhibitors of fatty acid synthase. J Med Chem51:5285-96 (2008) [PubMed] Article
Richardson, RD; Ma, G; Oyola, Y; Zancanella, M; Knowles, LM; Cieplak, P; Romo, D; Smith, JW Synthesis of novel beta-lactone inhibitors of fatty acid synthase. J Med Chem51:5285-96 (2008) [PubMed] Article