| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H4 receptor |
|---|
| Ligand | BDBM26391 |
|---|
| Substrate/Competitor | BDBM7966 |
|---|
| Meas. Tech. | Radioligand Binding Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 298.15±n/a K |
|---|
| Ki | 26±4 nM |
|---|
| Citation |  Cowart, MD; Altenbach, RJ; Liu, H; Hsieh, GC; Drizin, I; Milicic, I; Miller, TR; Witte, DG; Wishart, N; Fix-Stenzel, SR; McPherson, MJ; Adair, RM; Wetter, JM; Bettencourt, BM; Marsh, KC; Sullivan, JP; Honore, P; Esbenshade, TA; Brioni, JD Rotationally constrained 2,4-diamino-5,6-disubstituted pyrimidines: a new class of histamine H4 receptor antagonists with improved druglikeness and in vivo efficacy in pain and inflammation models. J Med Chem51:6547-57 (2008) [PubMed] Article Cowart, MD; Altenbach, RJ; Liu, H; Hsieh, GC; Drizin, I; Milicic, I; Miller, TR; Witte, DG; Wishart, N; Fix-Stenzel, SR; McPherson, MJ; Adair, RM; Wetter, JM; Bettencourt, BM; Marsh, KC; Sullivan, JP; Honore, P; Esbenshade, TA; Brioni, JD Rotationally constrained 2,4-diamino-5,6-disubstituted pyrimidines: a new class of histamine H4 receptor antagonists with improved druglikeness and in vivo efficacy in pain and inflammation models. J Med Chem51:6547-57 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Solution Info, Assay Method |
|---|
| |
| Histamine H4 receptor |
|---|
| Name: | Histamine H4 receptor |
|---|
| Synonyms: | AXOR35 | G-protein coupled receptor 105 | GPCR105 | GPRv53 | HH4R | HISTAMINE H4 | HRH4 | HRH4_HUMAN | Histamine H4 receptor | Histamine H4 receptor (H4R) | Histamine receptor (H3 and H4) | Pfi-013 | SP9144 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44517.02 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Binding assays were using CHO cells stably expressing hH4R receptors. |
|---|
| Residue: | 390 |
|---|
| Sequence: | MPDTNSTINLSLSTRVTLAFFMSLVAFAIMLGNALVILAFVVDKNLRHRSSYFFLNLAIS
DFFVGVISIPLYIPHTLFEWDFGKEICVFWLTTDYLLCTASVYNIVLISYDRYLSVSNAV
SYRTQHTGVLKIVTLMVAVWVLAFLVNGPMILVSESWKDEGSECEPGFFSEWYILAITSF
LEFVIPVILVAYFNMNIYWSLWKRDHLSRCQSHPGLTAVSSNICGHSFRGRLSSRRSLSA
STEVPASFHSERQRRKSSLMFSSRTKMNSNTIASKMGSFSQSDSVALHQREHVELLRARR
LAKSLAILLGVFAVCWAPYSLFTIVLSFYSSATGPKSVWYRIAFWLQWFNSFVNPLLYPL
CHKRFQKAFLKIFCIKKQPLPSQHSRSVSS
|
|
|
|---|
| BDBM26391 |
|---|
| BDBM7966 |
|---|
| Name | BDBM26391 |
|---|
| Synonyms: | 5-chloro-2-(piperazin-1-ylcarbonyl)-1H-indole | JNJ 7777120 analogue, 4 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H14ClN3O |
|---|
| Mol. Mass. | 263.723 |
|---|
| SMILES | Clc1ccc2[nH]c(cc2c1)C(=O)N1CCNCC1 |
|---|
| Structure | 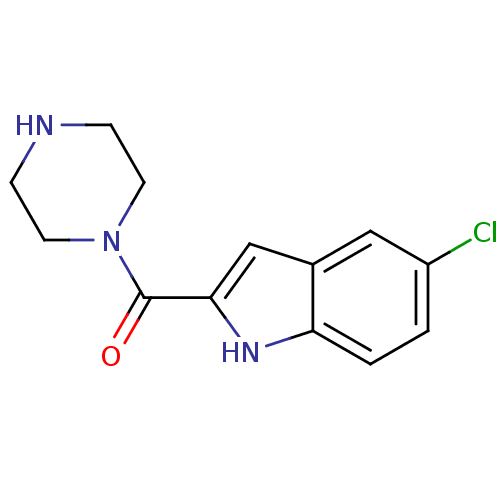
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cowart, MD; Altenbach, RJ; Liu, H; Hsieh, GC; Drizin, I; Milicic, I; Miller, TR; Witte, DG; Wishart, N; Fix-Stenzel, SR; McPherson, MJ; Adair, RM; Wetter, JM; Bettencourt, BM; Marsh, KC; Sullivan, JP; Honore, P; Esbenshade, TA; Brioni, JD Rotationally constrained 2,4-diamino-5,6-disubstituted pyrimidines: a new class of histamine H4 receptor antagonists with improved druglikeness and in vivo efficacy in pain and inflammation models. J Med Chem51:6547-57 (2008) [PubMed] Article
Cowart, MD; Altenbach, RJ; Liu, H; Hsieh, GC; Drizin, I; Milicic, I; Miller, TR; Witte, DG; Wishart, N; Fix-Stenzel, SR; McPherson, MJ; Adair, RM; Wetter, JM; Bettencourt, BM; Marsh, KC; Sullivan, JP; Honore, P; Esbenshade, TA; Brioni, JD Rotationally constrained 2,4-diamino-5,6-disubstituted pyrimidines: a new class of histamine H4 receptor antagonists with improved druglikeness and in vivo efficacy in pain and inflammation models. J Med Chem51:6547-57 (2008) [PubMed] Article