| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4/3A5 |
|---|
| Ligand | BDBM50305083 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | CYP Enzyme Inhibition Assay |
|---|
| pH | 7.4±n/a |
|---|
| Temperature | 310.15±n/a K |
|---|
| IC50 | >30000±0.0 nM |
|---|
| Comments | extracted |
|---|
| Citation |  Li, Y; Prakash, SR; Xia, CQ; Liao, MM Inhibitor of breast cancer resistance protein (BCRP) US Patent US9695174 Publication Date 7/4/2017 Li, Y; Prakash, SR; Xia, CQ; Liao, MM Inhibitor of breast cancer resistance protein (BCRP) US Patent US9695174 Publication Date 7/4/2017 |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4/3A5 |
|---|
| Name: | Cytochrome P450 3A4/3A5 |
|---|
| Synonyms: | CYP3A-M |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | n/a |
|---|
| Description: | n/a |
|---|
| Components: | This complex has 2 components. |
|---|
| Component 1 |
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| Component 2 |
| Name: | Cytochrome P450 3A5 |
|---|
| Synonyms: | CP3A5_HUMAN | CYP3A5 | Cytochrome P450 3A5 | Cytochrome P450 3A5 (CYP3A5) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 57118.00 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P20815 |
|---|
| Residue: | 502 |
|---|
| Sequence: | MDLIPNLAVETWLLLAVSLVLLYLYGTRTHGLFKRLGIPGPTPLPLLGNVLSYRQGLWKF
DTECYKKYGKMWGTYEGQLPVLAITDPDVIRTVLVKECYSVFTNRRSLGPVGFMKSAISL
AEDEEWKRIRSLLSPTFTSGKLKEMFPIIAQYGDVLVRNLRREAEKGKPVTLKDIFGAYS
MDVITGTSFGVNIDSLNNPQDPFVESTKKFLKFGFLDPLFLSIILFPFLTPVFEALNVSL
FPKDTINFLSKSVNRMKKSRLNDKQKHRLDFLQLMIDSQNSKETESHKALSDLELAAQSI
IFIFAGYETTSSVLSFTLYELATHPDVQQKLQKEIDAVLPNKAPPTYDAVVQMEYLDMVV
NETLRLFPVAIRLERTCKKDVEINGVFIPKGSMVVIPTYALHHDPKYWTEPEEFRPERFS
KKKDSIDPYIYTPFGTGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLDTQG
LLQPEKPIVLKVDSRDGTLSGE
|
|
|
|---|
| BDBM50305083 |
|---|
| n/a |
|---|
| Name | BDBM50305083 |
|---|
| Synonyms: | 3-((3S,6S)-6-Isobutyl-9-methoxy-1,4-dioxo-1,2,3,4,6,7,12,12a-octahydro-pyrazino[1',2':1,6]pyrido[3,4-b]indol-3-yl)-propionic acid tert-butyl ester | 3-((3S,6S,12aS)-6-Isobutyl-9-methoxy-1,4-dioxo-1,2,3,4,6,7,12,12a-octahydro-pyrazino[1',2':1,6]pyrido[3,4-b]indol-3-yl)-propionic acid tert-butyl ester | CHEMBL488910 | Ko-143 | Ko143 | US9695174, Ko143 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H35N3O5 |
|---|
| Mol. Mass. | 469.5732 |
|---|
| SMILES | COc1ccc2c3C[C@@H]4N([C@@H](CC(C)C)c3[nH]c2c1)C(=O)[C@H](CCC(=O)OC(C)(C)C)NC4=O |r| |
|---|
| Structure | 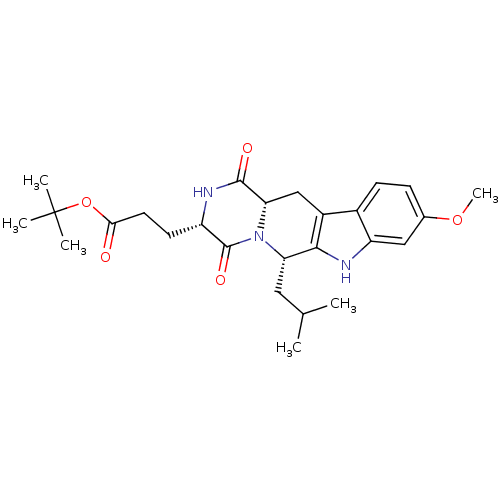
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, Y; Prakash, SR; Xia, CQ; Liao, MM Inhibitor of breast cancer resistance protein (BCRP) US Patent US9695174 Publication Date 7/4/2017
Li, Y; Prakash, SR; Xia, CQ; Liao, MM Inhibitor of breast cancer resistance protein (BCRP) US Patent US9695174 Publication Date 7/4/2017