| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 2 |
|---|
| Ligand | BDBM50244242 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1673538 (CHEMBL4023567) |
|---|
| Ki | 188±n/a nM |
|---|
| Citation |  Dolles, D; Hoffmann, M; Gunesch, S; Marinelli, O; M�ller, J; Santoni, G; Chatonnet, A; Lohse, MJ; Wittmann, HJ; Strasser, A; Nabissi, M; Maurice, T; Decker, M Structure-Activity Relationships and Computational Investigations into the Development of Potent and Balanced Dual-Acting Butyrylcholinesterase Inhibitors and Human Cannabinoid Receptor 2 Ligands with Pro-Cognitive in Vivo Profiles. J Med Chem61:1646-1663 (2018) [PubMed] Article Dolles, D; Hoffmann, M; Gunesch, S; Marinelli, O; M�ller, J; Santoni, G; Chatonnet, A; Lohse, MJ; Wittmann, HJ; Strasser, A; Nabissi, M; Maurice, T; Decker, M Structure-Activity Relationships and Computational Investigations into the Development of Potent and Balanced Dual-Acting Butyrylcholinesterase Inhibitors and Human Cannabinoid Receptor 2 Ligands with Pro-Cognitive in Vivo Profiles. J Med Chem61:1646-1663 (2018) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 2 |
|---|
| Name: | Cannabinoid receptor 2 |
|---|
| Synonyms: | CANNABINOID CB2 | CB-2 | CB2 | CB2A | CB2B | CNR2 | CNR2_HUMAN | CX5 | Cannabinoid CB2 receptor | Cannabinoid receptor 2 (CB2) | Cannabinoid receptor 2 (CB2R) | hCB2 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 39690.94 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P34972 |
|---|
| Residue: | 360 |
|---|
| Sequence: | MEECWVTEIANGSKDGLDSNPMKDYMILSGPQKTAVAVLCTLLGLLSALENVAVLYLILS
SHQLRRKPSYLFIGSLAGADFLASVVFACSFVNFHVFHGVDSKAVFLLKIGSVTMTFTAS
VGSLLLTAIDRYLCLRYPPSYKALLTRGRALVTLGIMWVLSALVSYLPLMGWTCCPRPCS
ELFPLIPNDYLLSWLLFIAFLFSGIIYTYGHVLWKAHQHVASLSGHQDRQVPGMARMRLD
VRLAKTLGLVLAVLLICWFPVLALMAHSLATTLSDQVKKAFAFCSMLCLINSMVNPVIYA
LRSGEIRSSAHHCLAHWKKCVRGLGSEAKEEAPRSSVTETEADGKITPWPDSRDLDLSDC
|
|
|
|---|
| BDBM50244242 |
|---|
| n/a |
|---|
| Name | BDBM50244242 |
|---|
| Synonyms: | CHEMBL4082936 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H38N4O2 |
|---|
| Mol. Mass. | 462.6269 |
|---|
| SMILES | CCOc1ccc(Cc2nc3cc(ccc3n2CCN2CCCCC2)C(=O)N(CC)CC)cc1 |
|---|
| Structure | 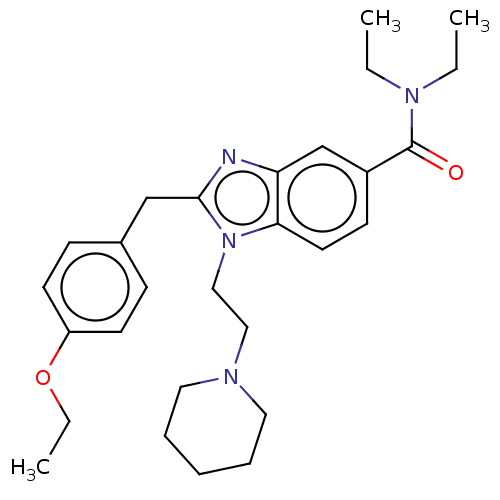
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Dolles, D; Hoffmann, M; Gunesch, S; Marinelli, O; M�ller, J; Santoni, G; Chatonnet, A; Lohse, MJ; Wittmann, HJ; Strasser, A; Nabissi, M; Maurice, T; Decker, M Structure-Activity Relationships and Computational Investigations into the Development of Potent and Balanced Dual-Acting Butyrylcholinesterase Inhibitors and Human Cannabinoid Receptor 2 Ligands with Pro-Cognitive in Vivo Profiles. J Med Chem61:1646-1663 (2018) [PubMed] Article
Dolles, D; Hoffmann, M; Gunesch, S; Marinelli, O; M�ller, J; Santoni, G; Chatonnet, A; Lohse, MJ; Wittmann, HJ; Strasser, A; Nabissi, M; Maurice, T; Decker, M Structure-Activity Relationships and Computational Investigations into the Development of Potent and Balanced Dual-Acting Butyrylcholinesterase Inhibitors and Human Cannabinoid Receptor 2 Ligands with Pro-Cognitive in Vivo Profiles. J Med Chem61:1646-1663 (2018) [PubMed] Article