| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 11B2, mitochondrial |
|---|
| Ligand | BDBM50249045 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1679698 (CHEMBL4029975) |
|---|
| IC50 | 14±n/a nM |
|---|
| Citation |  Hoyt, SB; Taylor, J; London, C; Ali, A; Ujjainwalla, F; Tata, J; Struthers, M; Cully, D; Wisniewski, T; Ren, N; Bopp, C; Sok, A; Verras, A; McMasters, D; Chen, Q; Tung, E; Tang, W; Salituro, G; Clemas, J; Zhou, G; MacNeil, D; Duffy, R; Xiong, Y Discovery of indazole aldosterone synthase (CYP11B2) inhibitors as potential treatments for hypertension. Bioorg Med Chem Lett27:2384-2388 (2017) [PubMed] Article Hoyt, SB; Taylor, J; London, C; Ali, A; Ujjainwalla, F; Tata, J; Struthers, M; Cully, D; Wisniewski, T; Ren, N; Bopp, C; Sok, A; Verras, A; McMasters, D; Chen, Q; Tung, E; Tang, W; Salituro, G; Clemas, J; Zhou, G; MacNeil, D; Duffy, R; Xiong, Y Discovery of indazole aldosterone synthase (CYP11B2) inhibitors as potential treatments for hypertension. Bioorg Med Chem Lett27:2384-2388 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 11B2, mitochondrial |
|---|
| Name: | Cytochrome P450 11B2, mitochondrial |
|---|
| Synonyms: | Aldosterone synthase | C11B2_RAT | CYPXIB2 | Cyp11b-2 | Cyp11b2 | Cytochrome P450 11B | Cytochrome P450 11B2 | Cytochrome P450 11B2, mitochondrial | P450-Aldo-1 | Steroid 11-beta-hydroxylase |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 58260.64 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_224979 |
|---|
| Residue: | 510 |
|---|
| Sequence: | MGACDNDFIELHSRVTADVWLARPWQCLHRTRALGTTATLAPKTLKPFEAIPQYSRNKWL
KMIQILREQGQENLHLEMHQAFQELGPIFRHSAGGAQIVSVMLPEDAEKLHQVESILPRR
MHLEPWVAHRELRGLRRGVFLLNGAEWRFNRLKLNPNVLSPKAVQNFVPMVDEVARDFLE
ALKKKVRQNARGSLTMDVQQSLFNYTIEASNFALFGERLGLLGHDLNPGSLKFIHALHSM
FKSTTQLLFLPRSLTRWTSTQVWKEHFDAWDVISEYANRCIWKVHQELRLGSSQTYSGIV
AALITQGALPLDAIKANSMELTAGSVDTTAIPLVMTLFELARNPDVQQALRQETLAAEAS
IAANPQKAMSDLPLLRAALKETLRLYPVGGFLERILNSDLVLQNYHVPAGTLVLLYLYSM
GRNPAVFPRPERYMPQRWLERKRSFQHLAFGFGVRQCLGRRLAEVEMLLLLHHMLKTFQV
ETLRQEDVQMAYRFVLMPSSSPVLTFRPIS
|
|
|
|---|
| BDBM50249045 |
|---|
| n/a |
|---|
| Name | BDBM50249045 |
|---|
| Synonyms: | CHEMBL4098297 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H14F3N3O |
|---|
| Mol. Mass. | 321.2971 |
|---|
| SMILES | Cc1nn(-c2cncc(c2)[C@](C)(O)C(F)(F)F)c2ccccc12 |r| |
|---|
| Structure | 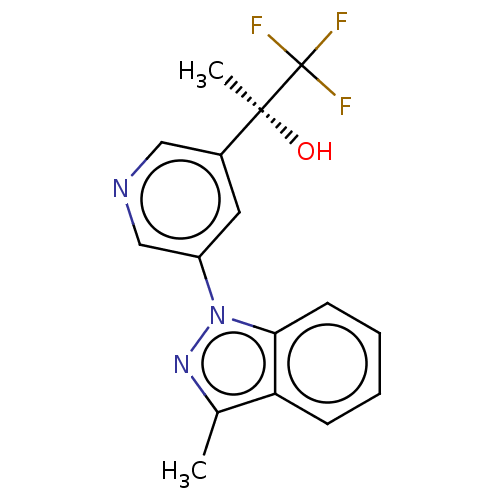
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hoyt, SB; Taylor, J; London, C; Ali, A; Ujjainwalla, F; Tata, J; Struthers, M; Cully, D; Wisniewski, T; Ren, N; Bopp, C; Sok, A; Verras, A; McMasters, D; Chen, Q; Tung, E; Tang, W; Salituro, G; Clemas, J; Zhou, G; MacNeil, D; Duffy, R; Xiong, Y Discovery of indazole aldosterone synthase (CYP11B2) inhibitors as potential treatments for hypertension. Bioorg Med Chem Lett27:2384-2388 (2017) [PubMed] Article
Hoyt, SB; Taylor, J; London, C; Ali, A; Ujjainwalla, F; Tata, J; Struthers, M; Cully, D; Wisniewski, T; Ren, N; Bopp, C; Sok, A; Verras, A; McMasters, D; Chen, Q; Tung, E; Tang, W; Salituro, G; Clemas, J; Zhou, G; MacNeil, D; Duffy, R; Xiong, Y Discovery of indazole aldosterone synthase (CYP11B2) inhibitors as potential treatments for hypertension. Bioorg Med Chem Lett27:2384-2388 (2017) [PubMed] Article