| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bile acid receptor |
|---|
| Ligand | BDBM50250869 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1682383 (CHEMBL4032660) |
|---|
| EC50 | 0.460000±n/a nM |
|---|
| Citation |  Tully, DC; Rucker, PV; Chianelli, D; Williams, J; Vidal, A; Alper, PB; Mutnick, D; Bursulaya, B; Schmeits, J; Wu, X; Bao, D; Zoll, J; Kim, Y; Groessl, T; McNamara, P; Seidel, HM; Molteni, V; Liu, B; Phimister, A; Joseph, SB; Laffitte, B Discovery of Tropifexor (LJN452), a Highly Potent Non-bile Acid FXR Agonist for the Treatment of Cholestatic Liver Diseases and Nonalcoholic Steatohepatitis (NASH). J Med Chem60:9960-9973 (2017) [PubMed] Article Tully, DC; Rucker, PV; Chianelli, D; Williams, J; Vidal, A; Alper, PB; Mutnick, D; Bursulaya, B; Schmeits, J; Wu, X; Bao, D; Zoll, J; Kim, Y; Groessl, T; McNamara, P; Seidel, HM; Molteni, V; Liu, B; Phimister, A; Joseph, SB; Laffitte, B Discovery of Tropifexor (LJN452), a Highly Potent Non-bile Acid FXR Agonist for the Treatment of Cholestatic Liver Diseases and Nonalcoholic Steatohepatitis (NASH). J Med Chem60:9960-9973 (2017) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bile acid receptor |
|---|
| Name: | Bile acid receptor |
|---|
| Synonyms: | BAR | Bile acid receptor FXR | FXR | Farnesol receptor HRR-1 | HRR1 | NR1H4 | NR1H4_HUMAN | Nuclear receptor subfamily 1 group H member 4 | RIP14 | RXR-interacting protein 14 | Retinoid X receptor-interacting protein 14 | farnesoid x receptor |
|---|
| Type: | Nuclear Receptor |
|---|
| Mol. Mass.: | 55916.24 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q96RI1 |
|---|
| Residue: | 486 |
|---|
| Sequence: | MVMQFQGLENPIQISPHCSCTPSGFFMEMMSMKPAKGVLTEQVAGPLGQNLEVEPYSQYS
NVQFPQVQPQISSSSYYSNLGFYPQQPEEWYSPGIYELRRMPAETLYQGETEVAEMPVTK
KPRMGASAGRIKGDELCVVCGDRASGYHYNALTCEGCKGFFRRSITKNAVYKCKNGGNCV
MDMYMRRKCQECRLRKCKEMGMLAECMYTGLLTEIQCKSKRLRKNVKQHADQTVNEDSEG
RDLRQVTSTTKSCREKTELTPDQQTLLHFIMDSYNKQRMPQEITNKILKEEFSAEENFLI
LTEMATNHVQVLVEFTKKLPGFQTLDHEDQIALLKGSAVEAMFLRSAEIFNKKLPSGHSD
LLEERIRNSGISDEYITPMFSFYKSIGELKMTQEEYALLTAIVILSPDRQYIKDREAVEK
LQEPLLDVLQKLCKIHQPENPQHFACLLGRLTELRTFNHHHAEMLMSWRVNDHKFTPLLC
EIWDVQ
|
|
|
|---|
| BDBM50250869 |
|---|
| n/a |
|---|
| Name | BDBM50250869 |
|---|
| Synonyms: | CHEMBL4061776 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H25F4N3O4S |
|---|
| Mol. Mass. | 587.585 |
|---|
| SMILES | OC(=O)c1cc(F)c2nc(sc2c1)N1C2CCC1CC(C2)OCc1c(onc1-c1ccccc1C(F)(F)F)C1CC1 |THB:9:13:15.16:18.19.20,21:19:13:15.16| |
|---|
| Structure | 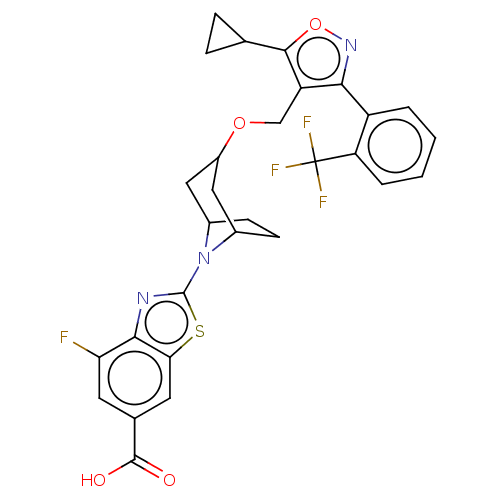
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Tully, DC; Rucker, PV; Chianelli, D; Williams, J; Vidal, A; Alper, PB; Mutnick, D; Bursulaya, B; Schmeits, J; Wu, X; Bao, D; Zoll, J; Kim, Y; Groessl, T; McNamara, P; Seidel, HM; Molteni, V; Liu, B; Phimister, A; Joseph, SB; Laffitte, B Discovery of Tropifexor (LJN452), a Highly Potent Non-bile Acid FXR Agonist for the Treatment of Cholestatic Liver Diseases and Nonalcoholic Steatohepatitis (NASH). J Med Chem60:9960-9973 (2017) [PubMed] Article
Tully, DC; Rucker, PV; Chianelli, D; Williams, J; Vidal, A; Alper, PB; Mutnick, D; Bursulaya, B; Schmeits, J; Wu, X; Bao, D; Zoll, J; Kim, Y; Groessl, T; McNamara, P; Seidel, HM; Molteni, V; Liu, B; Phimister, A; Joseph, SB; Laffitte, B Discovery of Tropifexor (LJN452), a Highly Potent Non-bile Acid FXR Agonist for the Treatment of Cholestatic Liver Diseases and Nonalcoholic Steatohepatitis (NASH). J Med Chem60:9960-9973 (2017) [PubMed] Article