| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50474742 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_159256 (CHEMBL764083) |
|---|
| IC50 | 177±n/a nM |
|---|
| Citation |  Shin, SS; Byun, Y; Lim, KM; Choi, JK; Lee, KW; Moh, JH; Kim, JK; Jeong, YS; Kim, JY; Choi, YH; Koh, HJ; Park, YH; Oh, YI; Noh, MS; Chung, S In vitro structure-activity relationship and in vivo studies for a novel class of cyclooxygenase-2 inhibitors: 5-aryl-2,2-dialkyl-4-phenyl-3(2H)furanone derivatives. J Med Chem47:792-804 (2004) [PubMed] Article Shin, SS; Byun, Y; Lim, KM; Choi, JK; Lee, KW; Moh, JH; Kim, JK; Jeong, YS; Kim, JY; Choi, YH; Koh, HJ; Park, YH; Oh, YI; Noh, MS; Chung, S In vitro structure-activity relationship and in vivo studies for a novel class of cyclooxygenase-2 inhibitors: 5-aryl-2,2-dialkyl-4-phenyl-3(2H)furanone derivatives. J Med Chem47:792-804 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | Cox-1 | Cox1 | Cyclooxygenase-1 | PGH synthase 1 | PGH1_MOUSE | PGHS-1 | PHS 1 | Prostaglandin G/H synthase (cyclooxygenase) | Prostaglandin H2 synthase 1 | Prostaglandin-endoperoxide synthase 1 | Ptgs1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 69044.61 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_10575 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MSRRSLSLWFPLLLLLLLPPTPSVLLADPGVPSPVNPCCYYPCQNQGVCVRFGLDNYQCD
CTRTGYSGPNCTIPEIWTWLRNSLRPSPSFTHFLLTHGYWLWEFVNATFIREVLMRLVLT
VRSNLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDVQLLA
QQLLLRREFIPAPQGTNILFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNL
ERQYHLRLFKDGKLKYQVLDGEVYPPSVEQASVLMRYPPGVPPERQMAVGQEVFGLLPGL
MLFSTIWLREHNRVCDLLKEEHPTWDDEQLFQTTRLILIGETIKIVIEEYVQHLSGYFLQ
LKFDPELLFRAQFQYRNRIAMEFNHLYHWHPLMPNSFQVGSQEYSYEQFLFNTSMLVDYG
VEALVDAFSRQRAGRIGGGRNFDYHVLHVAVDVIKESREMRLQPFNEYRKRFGLKPYTSF
QELTGEKEMAAELEELYGDIDALEFYPGLLLEKCQPNSIFGESMIEMGAPFSLKGLLGNP
ICSPEYWKPSTFGGDVGFNLVNTASLKKLVCLNTKTCPYVSFRVPDYPGDDGSVLVRRST
EL
|
|
|
|---|
| BDBM50474742 |
|---|
| n/a |
|---|
| Name | BDBM50474742 |
|---|
| Synonyms: | CHEMBL166295 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H17NO4S |
|---|
| Mol. Mass. | 343.397 |
|---|
| SMILES | CC1(C)OC(=C(C1=O)c1ccccc1)c1ccc(cc1)S(N)(=O)=O |c:4| |
|---|
| Structure | 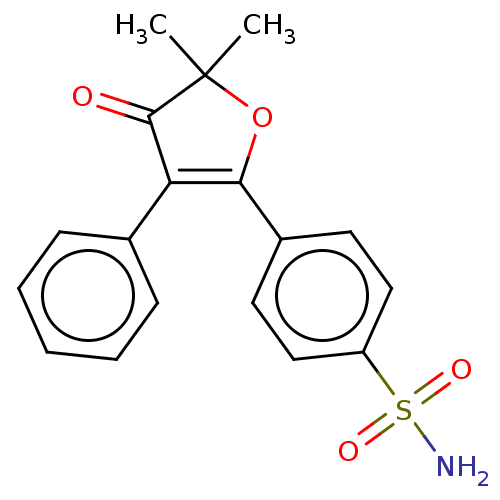
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Shin, SS; Byun, Y; Lim, KM; Choi, JK; Lee, KW; Moh, JH; Kim, JK; Jeong, YS; Kim, JY; Choi, YH; Koh, HJ; Park, YH; Oh, YI; Noh, MS; Chung, S In vitro structure-activity relationship and in vivo studies for a novel class of cyclooxygenase-2 inhibitors: 5-aryl-2,2-dialkyl-4-phenyl-3(2H)furanone derivatives. J Med Chem47:792-804 (2004) [PubMed] Article
Shin, SS; Byun, Y; Lim, KM; Choi, JK; Lee, KW; Moh, JH; Kim, JK; Jeong, YS; Kim, JY; Choi, YH; Koh, HJ; Park, YH; Oh, YI; Noh, MS; Chung, S In vitro structure-activity relationship and in vivo studies for a novel class of cyclooxygenase-2 inhibitors: 5-aryl-2,2-dialkyl-4-phenyl-3(2H)furanone derivatives. J Med Chem47:792-804 (2004) [PubMed] Article