| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Delta-type opioid receptor |
|---|
| Ligand | BDBM50068133 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_146652 (CHEMBL754936) |
|---|
| IC50 | 1.6±n/a nM |
|---|
| Citation |  Liao, S; Alfaro-Lopez, J; Shenderovich, MD; Hosohata, K; Lin, J; Li, X; Stropova, D; Davis, P; Jernigan, KA; Porreca, F; Yamamura, HI; Hruby, VJ De novo design, synthesis, and biological activities of high-affinity and selective non-peptide agonists of the delta-opioid receptor. J Med Chem41:4767-76 (1998) [PubMed] Article Liao, S; Alfaro-Lopez, J; Shenderovich, MD; Hosohata, K; Lin, J; Li, X; Stropova, D; Davis, P; Jernigan, KA; Porreca, F; Yamamura, HI; Hruby, VJ De novo design, synthesis, and biological activities of high-affinity and selective non-peptide agonists of the delta-opioid receptor. J Med Chem41:4767-76 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Delta-type opioid receptor |
|---|
| Name: | Delta-type opioid receptor |
|---|
| Synonyms: | Cytochrome P450 3A4 | DOR-1 | Delta opioid receptor | Delta-type opioid receptor | Delta-type opioid receptor (DOR) | OPIATE Delta | OPRD_RAT | Opiate Delta 1 | Opioid receptor | Opioid receptor A | Opioid receptors; mu & delta | Oprd1 | Ror-a | Voltage-gated potassium channel |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 40465.04 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Competition binding assays were using CHO-K1 cell membranes expressing the opioid receptor. |
|---|
| Residue: | 372 |
|---|
| Sequence: | MEPVPSARAELQFSLLANVSDTFPSAFPSASANASGSPGARSASSLALAIAITALYSAVC
AVGLLGNVLVMFGIVRYTKLKTATNIYIFNLALADALATSTLPFQSAKYLMETWPFGELL
CKAVLSIDYYNMFTSIFTLTMMSVDRYIAVCHPVKALDFRTPAKAKLINICIWVLASGVG
VPIMVMAVTQPRDGAVVCTLQFPSPSWYWDTVTKICVFLFAFVVPILIITVCYGLMLLRL
RSVRLLSGSKEKDRSLRRITRMVLVVVGAFVVCWAPIHIFVIVWTLVDINRRDPLVVAAL
HLCIALGYANSSLNPVLYAFLDENFKRCFRQLCRAPCGGQEPGSLRRPRQATARERVTAC
TPSDGPGGGAAA
|
|
|
|---|
| BDBM50068133 |
|---|
| n/a |
|---|
| Name | BDBM50068133 |
|---|
| Synonyms: | (4S,7S,13S)-13-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-7-(4-chloro-benzyl)-3,3,14,14-tetramethyl-6,9,12-trioxo-1,2-dithia-5,8,11-triaza-cyclotetradecane-4-carboxylic acid | CHEMBL143939 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C30H38ClN5O7S2 |
|---|
| Mol. Mass. | 680.235 |
|---|
| SMILES | CC1(C)SSC(C)(C)[C@@H](NC(=O)[C@@H](N)Cc2ccc(O)cc2)C(=O)NCC(=O)N[C@@H](Cc2ccc(Cl)cc2)C(=O)N[C@H]1C(O)=O |
|---|
| Structure | 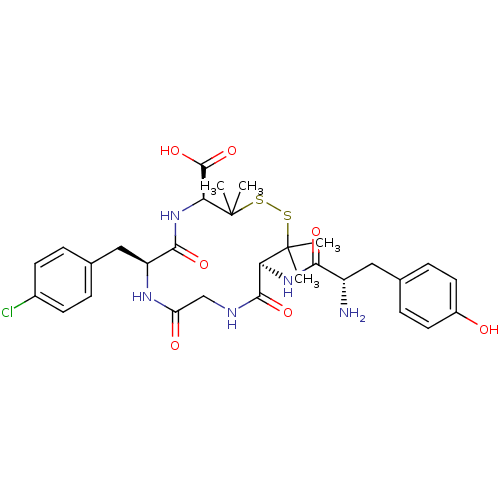
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Liao, S; Alfaro-Lopez, J; Shenderovich, MD; Hosohata, K; Lin, J; Li, X; Stropova, D; Davis, P; Jernigan, KA; Porreca, F; Yamamura, HI; Hruby, VJ De novo design, synthesis, and biological activities of high-affinity and selective non-peptide agonists of the delta-opioid receptor. J Med Chem41:4767-76 (1998) [PubMed] Article
Liao, S; Alfaro-Lopez, J; Shenderovich, MD; Hosohata, K; Lin, J; Li, X; Stropova, D; Davis, P; Jernigan, KA; Porreca, F; Yamamura, HI; Hruby, VJ De novo design, synthesis, and biological activities of high-affinity and selective non-peptide agonists of the delta-opioid receptor. J Med Chem41:4767-76 (1998) [PubMed] Article