| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histamine H3 receptor |
|---|
| Ligand | BDBM50483134 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1869141 (CHEMBL4370207) |
|---|
| Ki | 0.300000±n/a nM |
|---|
| Citation |  Nirogi, R; Shinde, A; Mohammed, AR; Badange, RK; Reballi, V; Bandyala, TR; Saraf, SK; Bojja, K; Manchineella, S; Achanta, PK; Kandukuri, KK; Subramanian, R; Benade, V; Palacharla, RC; Jayarajan, P; Pandey, S; Jasti, V Discovery and Development of N-[4-(1-Cyclobutylpiperidin-4-yloxy)phenyl]-2-(morpholin-4-yl)acetamide Dihydrochloride (SUVN-G3031): A Novel, Potent, Selective, and Orally Active Histamine H J Med Chem62:1203-1217 (2019) [PubMed] Article Nirogi, R; Shinde, A; Mohammed, AR; Badange, RK; Reballi, V; Bandyala, TR; Saraf, SK; Bojja, K; Manchineella, S; Achanta, PK; Kandukuri, KK; Subramanian, R; Benade, V; Palacharla, RC; Jayarajan, P; Pandey, S; Jasti, V Discovery and Development of N-[4-(1-Cyclobutylpiperidin-4-yloxy)phenyl]-2-(morpholin-4-yl)acetamide Dihydrochloride (SUVN-G3031): A Novel, Potent, Selective, and Orally Active Histamine H J Med Chem62:1203-1217 (2019) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histamine H3 receptor |
|---|
| Name: | Histamine H3 receptor |
|---|
| Synonyms: | G-protein coupled receptor 97 | GPCR97 | HH3R | HISTAMINE H3 | HRH3 | HRH3_HUMAN | Histamine H3 receptor (H3) | Histamine H3L | Histamine receptor (H3 and H4) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 48691.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Binding assays were using CHO cells stably expressing hH3R receptors. |
|---|
| Residue: | 445 |
|---|
| Sequence: | MERAPPDGPLNASGALAGEAAAAGGARGFSAAWTAVLAALMALLIVATVLGNALVMLAFV
ADSSLRTQNNFFLLNLAISDFLVGAFCIPLYVPYVLTGRWTFGRGLCKLWLVVDYLLCTS
SAFNIVLISYDRFLSVTRAVSYRAQQGDTRRAVRKMLLVWVLAFLLYGPAILSWEYLSGG
SSIPEGHCYAEFFYNWYFLITASTLEFFTPFLSVTFFNLSIYLNIQRRTRLRLDGAREAA
GPEPPPEAQPSPPPPPGCWGCWQKGHGEAMPLHRYGVGEAAVGAEAGEATLGGGGGGGSV
ASPTSSSGSSSRGTERPRSLKRGSKPSASSASLEKRMKMVSQSFTQRFRLSRDRKVAKSL
AVIVSIFGLCWAPYTLLMIIRAACHGHCVPDYWYETSFWLLWANSAVNPVLYPLCHHSFR
RAFTKLLCPQKLKIQPHSSLEHCWK
|
|
|
|---|
| BDBM50483134 |
|---|
| n/a |
|---|
| Name | BDBM50483134 |
|---|
| Synonyms: | CHEBI:64156 | Imetit |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C6H10N4S |
|---|
| Mol. Mass. | 170.235 |
|---|
| SMILES | NC(=N)SCCc1c[nH]cn1 |
|---|
| Structure | 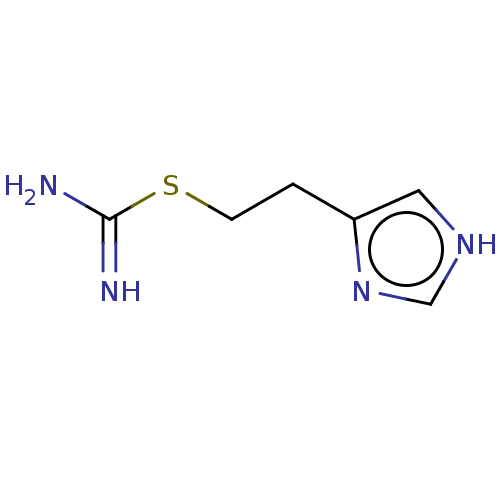
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nirogi, R; Shinde, A; Mohammed, AR; Badange, RK; Reballi, V; Bandyala, TR; Saraf, SK; Bojja, K; Manchineella, S; Achanta, PK; Kandukuri, KK; Subramanian, R; Benade, V; Palacharla, RC; Jayarajan, P; Pandey, S; Jasti, V Discovery and Development of N-[4-(1-Cyclobutylpiperidin-4-yloxy)phenyl]-2-(morpholin-4-yl)acetamide Dihydrochloride (SUVN-G3031): A Novel, Potent, Selective, and Orally Active Histamine H J Med Chem62:1203-1217 (2019) [PubMed] Article
Nirogi, R; Shinde, A; Mohammed, AR; Badange, RK; Reballi, V; Bandyala, TR; Saraf, SK; Bojja, K; Manchineella, S; Achanta, PK; Kandukuri, KK; Subramanian, R; Benade, V; Palacharla, RC; Jayarajan, P; Pandey, S; Jasti, V Discovery and Development of N-[4-(1-Cyclobutylpiperidin-4-yloxy)phenyl]-2-(morpholin-4-yl)acetamide Dihydrochloride (SUVN-G3031): A Novel, Potent, Selective, and Orally Active Histamine H J Med Chem62:1203-1217 (2019) [PubMed] Article