| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor alpha |
|---|
| Ligand | BDBM50085042 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_88427 (CHEMBL700787) |
|---|
| EC50 | 30000±n/a nM |
|---|
| Citation |  Willson, TM; Brown, PJ; Sternbach, DD; Henke, BR The PPARs: from orphan receptors to drug discovery. J Med Chem43:527-50 (2000) [PubMed] Willson, TM; Brown, PJ; Sternbach, DD; Henke, BR The PPARs: from orphan receptors to drug discovery. J Med Chem43:527-50 (2000) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor alpha |
|---|
| Name: | Peroxisome proliferator-activated receptor alpha |
|---|
| Synonyms: | NR1C1 | Nuclear receptor subfamily 1 group C member 1 | PPAR | PPAR alpha/gamma | PPAR-alpha | PPARA | PPARA_HUMAN | Peroxisome Proliferator-Activated Receptor alpha | Peroxisome proliferator-activated receptor | Peroxisome proliferator-activated receptor alpha (PPAR alpha) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52222.08 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q07869 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MVDTESPLCPLSPLEAGDLESPLSEEFLQEMGNIQEISQSIGEDSSGSFGFTEYQYLGSC
PGSDGSVITDTLSPASSPSSVTYPVVPGSVDESPSGALNIECRICGDKASGYHYGVHACE
GCKGFFRRTIRLKLVYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSE
KAKLKAEILTCEHDIEDSETADLKSLAKRIYEAYLKNFNMNKVKARVILSGKASNNPPFV
IHDMETLCMAEKTLVAKLVANGIQNKEAEVRIFHCCQCTSVETVTELTEFAKAIPGFANL
DLNDQVTLLKYGVYEAIFAMLSSVMNKDGMLVAYGNGFITREFLKSLRKPFCDIMEPKFD
FAMKFNALELDDSDISLFVAAIICCGDRPGLLNVGHIEKMQEGIVHVLRLHLQSNHPDDI
FLFPKLLQKMADLRQLVTEHAQLVQIIKKTESDAALHPLLQEIYRDMY
|
|
|
|---|
| BDBM50085042 |
|---|
| n/a |
|---|
| Name | BDBM50085042 |
|---|
| Synonyms: | 2-(4-(4-Chlorobenzoyl)phenoxy)-2-methylpropanoic acid 1-methylethyl ester | CHEMBL672 | FENOFIBRATE | FNF | Fenofibric acid | Finofibrate | Isopropyl (4'-(p-chlorobenzoyl)-2-phenoxy-2-methyl)propionate | Isopropyl 2-(4-(4-chlorobenzoyl)phenoxy)-2-methylpropionate | Lipantil (TN) | Procetofen | Tricor (TN) | propan-2-yl 2-[4-(4-chlorobenzoyl)phenoxy]-2-methylpropanoate |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H21ClO4 |
|---|
| Mol. Mass. | 360.831 |
|---|
| SMILES | CC(C)OC(=O)C(C)(C)Oc1ccc(cc1)C(=O)c1ccc(Cl)cc1 |
|---|
| Structure | 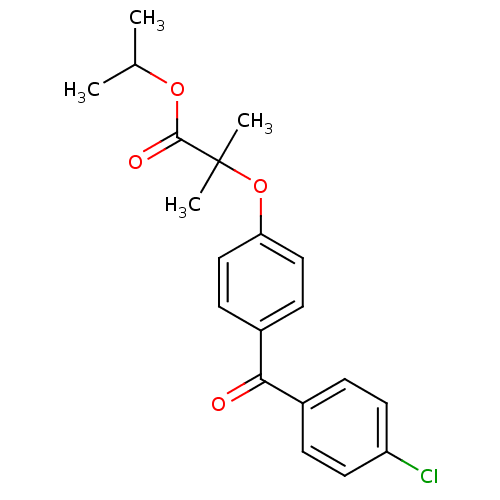
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Willson, TM; Brown, PJ; Sternbach, DD; Henke, BR The PPARs: from orphan receptors to drug discovery. J Med Chem43:527-50 (2000) [PubMed]
Willson, TM; Brown, PJ; Sternbach, DD; Henke, BR The PPARs: from orphan receptors to drug discovery. J Med Chem43:527-50 (2000) [PubMed]