| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Estrogen receptor |
|---|
| Ligand | BDBM50099587 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_99805 (CHEMBL710408) |
|---|
| IC50 | 1.5±n/a nM |
|---|
| Citation |  Miller, CP; Collini, MD; Tran, BD; Harris, HA; Kharode, YP; Marzolf, JT; Moran, RA; Henderson, RA; Bender, RH; Unwalla, RJ; Greenberger, LM; Yardley, JP; Abou-Gharbia, MA; Lyttle, CR; Komm, BS Design, synthesis, and preclinical characterization of novel, highly selective indole estrogens. J Med Chem44:1654-7 (2001) [PubMed] Miller, CP; Collini, MD; Tran, BD; Harris, HA; Kharode, YP; Marzolf, JT; Moran, RA; Henderson, RA; Bender, RH; Unwalla, RJ; Greenberger, LM; Yardley, JP; Abou-Gharbia, MA; Lyttle, CR; Komm, BS Design, synthesis, and preclinical characterization of novel, highly selective indole estrogens. J Med Chem44:1654-7 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Estrogen receptor |
|---|
| Name: | Estrogen receptor |
|---|
| Synonyms: | ER | ER-alpha | ESR | ESR1 | ESR1_HUMAN | Estradiol receptor | Estrogen receptor | Estrogen receptor (ER alpha) | Estrogen receptor (ER-alpha) | Estrogen receptor alpha (ER alpha) | Estrogen receptor alpha (ER) | NR3A1 | Nuclear receptor subfamily 3 group A member 1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 66230.44 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P03372 |
|---|
| Residue: | 595 |
|---|
| Sequence: | MTMTLHTKASGMALLHQIQGNELEPLNRPQLKIPLERPLGEVYLDSSKPAVYNYPEGAAY
EFNAAAAANAQVYGQTGLPYGPGSEAAAFGSNGLGGFPPLNSVSPSPLMLLHPPPQLSPF
LQPHGQQVPYYLENEPSGYTVREAGPPAFYRPNSDNRRQGGRERLASTNDKGSMAMESAK
ETRYCAVCNDYASGYHYGVWSCEGCKAFFKRSIQGHNDYMCPATNQCTIDKNRRKSCQAC
RLRKCYEVGMMKGGIRKDRRGGRMLKHKRQRDDGEGRGEVGSAGDMRAANLWPSPLMIKR
SKKNSLALSLTADQMVSALLDAEPPILYSEYDPTRPFSEASMMGLLTNLADRELVHMINW
AKRVPGFVDLTLHDQVHLLECAWLEILMIGLVWRSMEHPGKLLFAPNLLLDRNQGKCVEG
MVEIFDMLLATSSRFRMMNLQGEEFVCLKSIILLNSGVYTFLSSTLKSLEEKDHIHRVLD
KITDTLIHLMAKAGLTLQQQHQRLAQLLLILSHIRHMSNKGMEHLYSMKCKNVVPLYDLL
LEMLDAHRLHAPTSRGGASVEETDQSHLATAGSTSSHSLQKYYITGEAEGFPATV
|
|
|
|---|
| BDBM50099587 |
|---|
| n/a |
|---|
| Name | BDBM50099587 |
|---|
| Synonyms: | 2-(4-Hydroxy-phenyl)-3-methyl-1-[4-(2-piperidin-1-yl-ethoxy)-benzyl]-1H-indol-5-ol | CHEMBL44426 | Pipendoxifene | US8815934, No. 107 | US8815934, No. 97 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H32N2O3 |
|---|
| Mol. Mass. | 456.576 |
|---|
| SMILES | Cc1c(-c2ccc(O)cc2)n(Cc2ccc(OCCN3CCCCC3)cc2)c2ccc(O)cc12 |
|---|
| Structure | 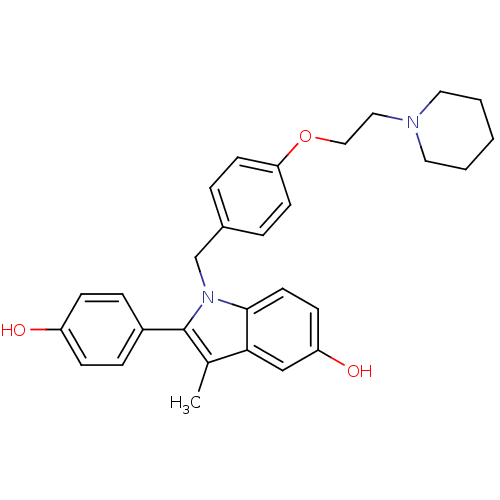
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Miller, CP; Collini, MD; Tran, BD; Harris, HA; Kharode, YP; Marzolf, JT; Moran, RA; Henderson, RA; Bender, RH; Unwalla, RJ; Greenberger, LM; Yardley, JP; Abou-Gharbia, MA; Lyttle, CR; Komm, BS Design, synthesis, and preclinical characterization of novel, highly selective indole estrogens. J Med Chem44:1654-7 (2001) [PubMed]
Miller, CP; Collini, MD; Tran, BD; Harris, HA; Kharode, YP; Marzolf, JT; Moran, RA; Henderson, RA; Bender, RH; Unwalla, RJ; Greenberger, LM; Yardley, JP; Abou-Gharbia, MA; Lyttle, CR; Komm, BS Design, synthesis, and preclinical characterization of novel, highly selective indole estrogens. J Med Chem44:1654-7 (2001) [PubMed]