| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 1A2 |
|---|
| Ligand | BDBM50545576 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1996019 (CHEMBL4629914) |
|---|
| IC50 | >20000±n/a nM |
|---|
| Citation |  Fromont, C; Atzori, A; Kaur, D; Hashmi, L; Greco, G; Cabanillas, A; Nguyen, HV; Jones, DH; Garz�n, M; Varela, A; Stevenson, B; Iacobini, GP; Lenoir, M; Rajesh, S; Box, C; Kumar, J; Grant, P; Novitskaya, V; Morgan, J; Sorrell, FJ; Redondo, C; Kramer, A; Harris, CJ; Leighton, B; Vickers, SP; Cheetham, SC; Kenyon, C; Grabowska, AM; Overduin, M; Berditchevski, F; Weston, CJ; Knapp, S; Fischer, PM; Butterworth, S Discovery of Highly Selective Inhibitors of Calmodulin-Dependent Kinases That Restore Insulin Sensitivity in the Diet-Induced Obesity J Med Chem63:6784-6801 (2020) [PubMed] Article Fromont, C; Atzori, A; Kaur, D; Hashmi, L; Greco, G; Cabanillas, A; Nguyen, HV; Jones, DH; Garz�n, M; Varela, A; Stevenson, B; Iacobini, GP; Lenoir, M; Rajesh, S; Box, C; Kumar, J; Grant, P; Novitskaya, V; Morgan, J; Sorrell, FJ; Redondo, C; Kramer, A; Harris, CJ; Leighton, B; Vickers, SP; Cheetham, SC; Kenyon, C; Grabowska, AM; Overduin, M; Berditchevski, F; Weston, CJ; Knapp, S; Fischer, PM; Butterworth, S Discovery of Highly Selective Inhibitors of Calmodulin-Dependent Kinases That Restore Insulin Sensitivity in the Diet-Induced Obesity J Med Chem63:6784-6801 (2020) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 1A2 |
|---|
| Name: | Cytochrome P450 1A2 |
|---|
| Synonyms: | CP1A2_HUMAN | CYP1A2 | CYPIA2 | Cholesterol 25-hydroxylase | Cytochrome P(3)450 | Cytochrome P450 1A | Cytochrome P450 1A2 (CYP1A2) | Cytochrome P450 4 | Cytochrome P450-P3 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 58423.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05177 |
|---|
| Residue: | 516 |
|---|
| Sequence: | MALSQSVPFSATELLLASAIFCLVFWVLKGLRPRVPKGLKSPPEPWGWPLLGHVLTLGKN
PHLALSRMSQRYGDVLQIRIGSTPVLVLSRLDTIRQALVRQGDDFKGRPDLYTSTLITDG
QSLTFSTDSGPVWAARRRLAQNALNTFSIASDPASSSSCYLEEHVSKEAKALISRLQELM
AGPGHFDPYNQVVVSVANVIGAMCFGQHFPESSDEMLSLVKNTHEFVETASSGNPLDFFP
ILRYLPNPALQRFKAFNQRFLWFLQKTVQEHYQDFDKNSVRDITGALFKHSKKGPRASGN
LIPQEKIVNLVNDIFGAGFDTVTTAISWSLMYLVTKPEIQRKIQKELDTVIGRERRPRLS
DRPQLPYLEAFILETFRHSSFLPFTIPHSTTRDTTLNGFYIPKKCCVFVNQWQVNHDPEL
WEDPSEFRPERFLTADGTAINKPLSEKMMLFGMGKRRCIGEVLAKWEIFLFLAILLQQLE
FSVPPGVKVDLTPIYGLTMKHARCEHVQARLRFSIN
|
|
|
|---|
| BDBM50545576 |
|---|
| n/a |
|---|
| Name | BDBM50545576 |
|---|
| Synonyms: | CHEMBL4640712 | US11530193, Example 50 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H30N8O |
|---|
| Mol. Mass. | 446.548 |
|---|
| SMILES | CC(C)(C#N)c1cc(Nc2nc(ncc2C(N)=O)N2CCC[C@H](N)C2)cc(c1)C(C)(C)C#N |r| |
|---|
| Structure | 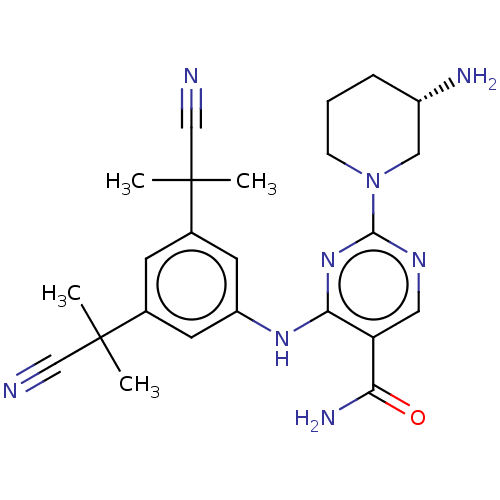
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fromont, C; Atzori, A; Kaur, D; Hashmi, L; Greco, G; Cabanillas, A; Nguyen, HV; Jones, DH; Garz�n, M; Varela, A; Stevenson, B; Iacobini, GP; Lenoir, M; Rajesh, S; Box, C; Kumar, J; Grant, P; Novitskaya, V; Morgan, J; Sorrell, FJ; Redondo, C; Kramer, A; Harris, CJ; Leighton, B; Vickers, SP; Cheetham, SC; Kenyon, C; Grabowska, AM; Overduin, M; Berditchevski, F; Weston, CJ; Knapp, S; Fischer, PM; Butterworth, S Discovery of Highly Selective Inhibitors of Calmodulin-Dependent Kinases That Restore Insulin Sensitivity in the Diet-Induced Obesity J Med Chem63:6784-6801 (2020) [PubMed] Article
Fromont, C; Atzori, A; Kaur, D; Hashmi, L; Greco, G; Cabanillas, A; Nguyen, HV; Jones, DH; Garz�n, M; Varela, A; Stevenson, B; Iacobini, GP; Lenoir, M; Rajesh, S; Box, C; Kumar, J; Grant, P; Novitskaya, V; Morgan, J; Sorrell, FJ; Redondo, C; Kramer, A; Harris, CJ; Leighton, B; Vickers, SP; Cheetham, SC; Kenyon, C; Grabowska, AM; Overduin, M; Berditchevski, F; Weston, CJ; Knapp, S; Fischer, PM; Butterworth, S Discovery of Highly Selective Inhibitors of Calmodulin-Dependent Kinases That Restore Insulin Sensitivity in the Diet-Induced Obesity J Med Chem63:6784-6801 (2020) [PubMed] Article