| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Genome polyprotein |
|---|
| Ligand | BDBM50110002 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_143628 |
|---|
| IC50 | 42±n/a nM |
|---|
| Citation |  Beevers, R; Carr, MG; Jones, PS; Jordan, S; Kay, PB; Lazell, RC; Raynham, TM Solution and solid-phase synthesis of potent inhibitors of hepatitis C virus NS3 proteinase. Bioorg Med Chem Lett12:641-3 (2002) [PubMed] Beevers, R; Carr, MG; Jones, PS; Jordan, S; Kay, PB; Lazell, RC; Raynham, TM Solution and solid-phase synthesis of potent inhibitors of hepatitis C virus NS3 proteinase. Bioorg Med Chem Lett12:641-3 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Genome polyprotein |
|---|
| Name: | Genome polyprotein |
|---|
| Synonyms: | Hepatitis C virus NS3 protease/helicase | Hepatitis C virus serine protease, NS3/NS4A |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 67067.41 |
|---|
| Organism: | Hepatitis C virus |
|---|
| Description: | A3EZI9 |
|---|
| Residue: | 631 |
|---|
| Sequence: | APITAYAQQTRGLLGCIITSLTGRDKNQVEGEVQIVSTAAQTFLATCINGVCWTVYHGAG
TRTIASSKGPVIQMYTNVDQDLVGWPAPQGARSLTPCTCGSSDLYLVTRHADVIPVRRRG
DGRGSLLSPRPISYLKGSSGGPLLCPAGHAVGIFRAAVCTRGVAKAVDFIPVEGLETTMR
SPVFSDNSSPPAVPQSYQVAHLHAPTGSGKSTKVPAAYAAQGYKVLVLNPSVAATLGFGA
YMSKAHGIDPNIRTGVRTITTGSPITYSTYGKFLADGGCSGGAYDIIICDECHSTDATSI
LGIGTVLDQAETAGARLTVLATATPPGSVTVPHPNIEEVALSTTGEIPFYGKAIPLEAIK
GGRHLIFCHSKKKCDELAAKLVALGVNAVAYYRGLDVSVIPASGDVVVVATDALMTGFTG
DFDSVIDCNTCVTQTVDFSLDPTFTIETTTLPQDAVSRTQRRGRTGRGKPGIYRFVTPGE
RPSGMFDSSVLCECYDAGCAWYELTPAETTVRLRAYMNTPGLPVCQDHLEFWEGVFTGLT
HIDAHFLSQTKQSGENLPYLVAYQATVCARAQAPPPSWDQMWKCLIRLKPTLHGPTPLLY
RLGAVQNEITLTHPITKYIMTCMSADLEVVT
|
|
|
|---|
| BDBM50110002 |
|---|
| n/a |
|---|
| Name | BDBM50110002 |
|---|
| Synonyms: | (S)-4-[(S)-1-({[(S)-1-((S)-1-Aminooxalyl-pentylcarbamoyl)-3-methyl-butylcarbamoyl]-phenyl-methyl}-carbamoyl)-2-o-tolyl-ethylcarbamoyl]-4-[(S)-3-carboxy-2-(3-carboxy-propionylamino)-propionylamino]-butyric acid | CHEMBL351341 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C44H59N7O14 |
|---|
| Mol. Mass. | 909.9778 |
|---|
| SMILES | CCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](Cc1ccccc1C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CCC(O)=O)c1ccccc1)C(=O)C(N)=O |
|---|
| Structure | 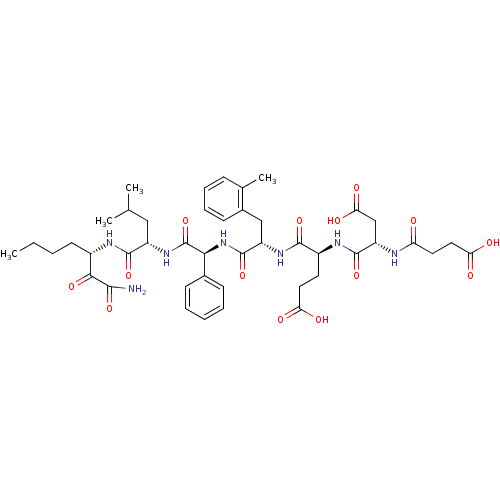
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Beevers, R; Carr, MG; Jones, PS; Jordan, S; Kay, PB; Lazell, RC; Raynham, TM Solution and solid-phase synthesis of potent inhibitors of hepatitis C virus NS3 proteinase. Bioorg Med Chem Lett12:641-3 (2002) [PubMed]
Beevers, R; Carr, MG; Jones, PS; Jordan, S; Kay, PB; Lazell, RC; Raynham, TM Solution and solid-phase synthesis of potent inhibitors of hepatitis C virus NS3 proteinase. Bioorg Med Chem Lett12:641-3 (2002) [PubMed]