| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Bifunctional purine biosynthesis protein ATIC |
|---|
| Ligand | BDBM66082 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2059607 (CHEMBL4714608) |
|---|
| Ki | 880±n/a nM |
|---|
| Citation |  Wallace-Povirk, A; Tong, N; Wong-Roushar, J; O'Connor, C; Zhou, X; Hou, Z; Bao, X; Garcia, GE; Li, J; Kim, S; Dann, CE; Matherly, LH; Gangjee, A Discovery of 6-substituted thieno[2,3-d]pyrimidine analogs as dual inhibitors of glycinamide ribonucleotide formyltransferase and 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase in de novo purine nucleotide biosynthesis in folate receptor expressing human tumors. Bioorg Med Chem37:0 (2021) [PubMed] Article Wallace-Povirk, A; Tong, N; Wong-Roushar, J; O'Connor, C; Zhou, X; Hou, Z; Bao, X; Garcia, GE; Li, J; Kim, S; Dann, CE; Matherly, LH; Gangjee, A Discovery of 6-substituted thieno[2,3-d]pyrimidine analogs as dual inhibitors of glycinamide ribonucleotide formyltransferase and 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase in de novo purine nucleotide biosynthesis in folate receptor expressing human tumors. Bioorg Med Chem37:0 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Bifunctional purine biosynthesis protein ATIC |
|---|
| Name: | Bifunctional purine biosynthesis protein ATIC |
|---|
| Synonyms: | 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase | 5-aminoimidazole-4-carboxamide-ribonucleotide transformylase | AICAR Tfase | AICAR transformylase | ATIC | Aminoimidazole carboxamide ribonucleotide transformylase (AICAR Tfase) | Bifunctional purine biosynthesis protein PURH | IMP Cyclohydrolase (IMPCH) | IMP cyclohydrolase | IMP synthetase | Inosinicase | PUR9_HUMAN | PURH | Phosphoribosylaminoimidazolecarboxamide formyltransferase | Thymidylate synthase/GAR transformylase/AICAR transformylase |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 64616.62 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P31939 |
|---|
| Residue: | 592 |
|---|
| Sequence: | MAPGQLALFSVSDKTGLVEFARNLTALGLNLVASGGTAKALRDAGLAVRDVSELTGFPEM
LGGRVKTLHPAVHAGILARNIPEDNADMARLDFNLIRVVACNLYPFVKTVASPGVTVEEA
VEQIDIGGVTLLRAAAKNHARVTVVCEPEDYVVVSTEMQSSESKDTSLETRRQLALKAFT
HTAQYDEAISDYFRKQYSKGVSQMPLRYGMNPHQTPAQLYTLQPKLPITVLNGAPGFINL
CDALNAWQLVKELKEALGIPAAASFKHVSPAGAAVGIPLSEDEAKVCMVYDLYKTLTPIS
AAYARARGADRMSSFGDFVALSDVCDVPTAKIISREVSDGIIAPGYEEEALTILSKKKNG
NYCVLQMDQSYKPDENEVRTLFGLHLSQKRNNGVVDKSLFSNVVTKNKDLPESALRDLIV
ATIAVKYTQSNSVCYAKNGQVIGIGAGQQSRIHCTRLAGDKANYWWLRHHPQVLSMKFKT
GVKRAEISNAIDQYVTGTIGEDEDLIKWKALFEEVPELLTEAEKKEWVEKLTEVSISSDA
FFPFRDNVDRAKRSGVAYIAAPSGSAADKVVIEACDELGIILAHTNLRLFHH
|
|
|
|---|
| BDBM66082 |
|---|
| n/a |
|---|
| Name | BDBM66082 |
|---|
| Synonyms: | (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methyl-amino]benzoyl]amino]glutaric acid;hydrate | (2S)-2-[[4-[(2,4-diaminopteridin-6-yl)methyl-methylamino]benzoyl]amino]pentanedioic acid;hydrate | (2S)-2-[[4-[[2,4-bis(azanyl)pteridin-6-yl]methyl-methyl-amino]phenyl]carbonylamino]pentanedioic acid;hydrate | (2S)-2-[[[4-[(2,4-diamino-6-pteridinyl)methyl-methylamino]phenyl]-oxomethyl]amino]pentanedioic acid;hydrate | L-Glutamic acid, N-[4-[[(2,4-diamino-6-pteridinyl)methyl]methylamino]benzoyl]- [CAS] | MLS001401431 | SMR000449324 | US11530198, Example Methotrexate | cid_165528 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H22N8O5 |
|---|
| Mol. Mass. | 454.4393 |
|---|
| SMILES | CN(Cc1cnc2nc(N)nc(N)c2n1)c1ccc(cc1)C(=O)N[C@@H](CCC(O)=O)C(O)=O |
|---|
| Structure | 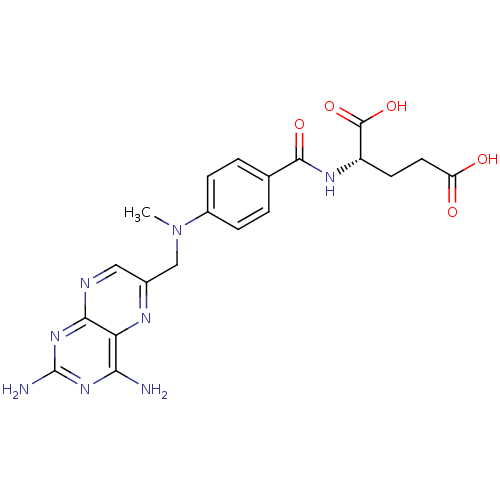
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Wallace-Povirk, A; Tong, N; Wong-Roushar, J; O'Connor, C; Zhou, X; Hou, Z; Bao, X; Garcia, GE; Li, J; Kim, S; Dann, CE; Matherly, LH; Gangjee, A Discovery of 6-substituted thieno[2,3-d]pyrimidine analogs as dual inhibitors of glycinamide ribonucleotide formyltransferase and 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase in de novo purine nucleotide biosynthesis in folate receptor expressing human tumors. Bioorg Med Chem37:0 (2021) [PubMed] Article
Wallace-Povirk, A; Tong, N; Wong-Roushar, J; O'Connor, C; Zhou, X; Hou, Z; Bao, X; Garcia, GE; Li, J; Kim, S; Dann, CE; Matherly, LH; Gangjee, A Discovery of 6-substituted thieno[2,3-d]pyrimidine analogs as dual inhibitors of glycinamide ribonucleotide formyltransferase and 5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase in de novo purine nucleotide biosynthesis in folate receptor expressing human tumors. Bioorg Med Chem37:0 (2021) [PubMed] Article