| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Ephrin type-A receptor 2 |
|---|
| Ligand | BDBM50571440 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2114669 (CHEMBL4823610) |
|---|
| IC50 | >100±n/a nM |
|---|
| Citation |  Zhang, H; Peng, X; Dai, Y; Shao, J; Ji, Y; Sun, Y; Liu, B; Cheng, X; Ai, J; Duan, W Discovery of a Pyrimidinedione Derivative as a Potent and Orally Bioavailable Axl Inhibitor. J Med Chem64:3956-3975 (2021) [PubMed] Article Zhang, H; Peng, X; Dai, Y; Shao, J; Ji, Y; Sun, Y; Liu, B; Cheng, X; Ai, J; Duan, W Discovery of a Pyrimidinedione Derivative as a Potent and Orally Bioavailable Axl Inhibitor. J Med Chem64:3956-3975 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Ephrin type-A receptor 2 |
|---|
| Name: | Ephrin type-A receptor 2 |
|---|
| Synonyms: | ECK | EPHA2 | EPHA2_HUMAN | Ephrin receptor | Epithelial cell kinase | Tyrosine-protein kinase receptor ECK |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 108260.70 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1505248 |
|---|
| Residue: | 976 |
|---|
| Sequence: | MELQAARACFALLWGCALAAAAAAQGKEVVLLDFAAAGGELGWLTHPYGKGWDLMQNIMN
DMPIYMYSVCNVMSGDQDNWLRTNWVYRGEAERIFIELKFTVRDCNSFPGGASSCKETFN
LYYAESDLDYGTNFQKRLFTKIDTIAPDEITVSSDFEARHVKLNVEERSVGPLTRKGFYL
AFQDIGACVALLSVRVYYKKCPELLQGLAHFPETIAGSDAPSLATVAGTCVDHAVVPPGG
EEPRMHCAVDGEWLVPIGQCLCQAGYEKVEDACQACSPGFFKFEASESPCLECPEHTLPS
PEGATSCECEEGFFRAPQDPASMPCTRPPSAPHYLTAVGMGAKVELRWTPPQDSGGREDI
VYSVTCEQCWPESGECGPCEASVRYSEPPHGLTRTSVTVSDLEPHMNYTFTVEARNGVSG
LVTSRSFRTASVSINQTEPPKVRLEGRSTTSLSVSWSIPPPQQSRVWKYEVTYRKKGDSN
SYNVRRTEGFSVTLDDLAPDTTYLVQVQALTQEGQGAGSKVHEFQTLSPEGSGNLAVIGG
VAVGVVLLLVLAGVGFFIHRRRKNQRARQSPEDVYFSKSEQLKPLKTYVDPHTYEDPNQA
VLKFTTEIHPSCVTRQKVIGAGEFGEVYKGMLKTSSGKKEVPVAIKTLKAGYTEKQRVDF
LGEAGIMGQFSHHNIIRLEGVISKYKPMMIITEYMENGALDKFLREKDGEFSVLQLVGML
RGIAAGMKYLANMNYVHRDLAARNILVNSNLVCKVSDFGLSRVLEDDPEATYTTSGGKIP
IRWTAPEAISYRKFTSASDVWSFGIVMWEVMTYGERPYWELSNHEVMKAINDGFRLPTPM
DCPSAIYQLMMQCWQQERARRPKFADIVSILDKLIRAPDSLKTLADFDPRVSIRLPSTSG
SEGVPFRTVSEWLESIKMQQYTEHFMAAGYTAIEKVVQMTNDDIKRIGVRLPGHQKRIAY
SLLGLKDQVNTVGIPI
|
|
|
|---|
| BDBM50571440 |
|---|
| n/a |
|---|
| Name | BDBM50571440 |
|---|
| Synonyms: | CHEMBL4873571 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H25F2N5O5 |
|---|
| Mol. Mass. | 561.5361 |
|---|
| SMILES | CC(C)n1cc(C(=O)Nc2ccc(Oc3ccnc(NC(=O)C4CC4)c3)c(F)c2)c(=O)n(-c2ccc(F)cc2)c1=O |
|---|
| Structure | 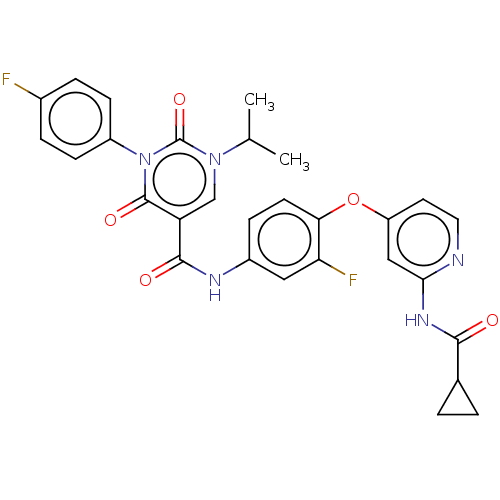
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, H; Peng, X; Dai, Y; Shao, J; Ji, Y; Sun, Y; Liu, B; Cheng, X; Ai, J; Duan, W Discovery of a Pyrimidinedione Derivative as a Potent and Orally Bioavailable Axl Inhibitor. J Med Chem64:3956-3975 (2021) [PubMed] Article
Zhang, H; Peng, X; Dai, Y; Shao, J; Ji, Y; Sun, Y; Liu, B; Cheng, X; Ai, J; Duan, W Discovery of a Pyrimidinedione Derivative as a Potent and Orally Bioavailable Axl Inhibitor. J Med Chem64:3956-3975 (2021) [PubMed] Article