| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Menin |
|---|
| Ligand | BDBM50582738 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2155531 (CHEMBL5040191) |
|---|
| IC50 | 2.8±n/a nM |
|---|
| Citation |  Zhang, M; Aguilar, A; Xu, S; Huang, L; Chinnaswamy, K; Sleger, T; Wang, B; Gross, S; Nicolay, BN; Ronseaux, S; Harvey, K; Wang, Y; McEachern, D; Kirchhoff, PD; Liu, Z; Stuckey, J; Tron, AE; Liu, T; Wang, S Discovery of M-1121 as an Orally Active Covalent Inhibitor of Menin-MLL Interaction Capable of Achieving Complete and Long-Lasting Tumor Regression. J Med Chem64:10333-10349 (2021) [PubMed] Article Zhang, M; Aguilar, A; Xu, S; Huang, L; Chinnaswamy, K; Sleger, T; Wang, B; Gross, S; Nicolay, BN; Ronseaux, S; Harvey, K; Wang, Y; McEachern, D; Kirchhoff, PD; Liu, Z; Stuckey, J; Tron, AE; Liu, T; Wang, S Discovery of M-1121 as an Orally Active Covalent Inhibitor of Menin-MLL Interaction Capable of Achieving Complete and Long-Lasting Tumor Regression. J Med Chem64:10333-10349 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Menin |
|---|
| Name: | Menin |
|---|
| Synonyms: | MEN1 | MEN1_HUMAN | Menin/Histone-lysine N-methyltransferase MLL | SCG2 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 68022.55 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | O00255 |
|---|
| Residue: | 610 |
|---|
| Sequence: | MGLKAAQKTLFPLRSIDDVVRLFAAELGREEPDLVLLSLVLGFVEHFLAVNRVIPTNVPE
LTFQPSPAPDPPGGLTYFPVADLSIIAALYARFTAQIRGAVDLSLYPREGGVSSRELVKK
VSDVIWNSLSRSYFKDRAHIQSLFSFITGTKLDSSGVAFAVVGACQALGLRDVHLALSED
HAWVVFGPNGEQTAEVTWHGKGNEDRRGQTVNAGVAERSWLYLKGSYMRCDRKMEVAFMV
CAINPSIDLHTDSLELLQLQQKLLWLLYDLGHLERYPMALGNLADLEELEPTPGRPDPLT
LYHKGIASAKTYYRDEHIYPYMYLAGYHCRNRNVREALQAWADTATVIQDYNYCREDEEI
YKEFFEVANDVIPNLLKEAASLLEAGEERPGEQSQGTQSQGSALQDPECFAHLLRFYDGI
CKWEEGSPTPVLHVGWATFLVQSLGRFEGQVRQKVRIVSREAEAAEAEEPWGEEAREGRR
RGPRRESKPEEPPPPKKPALDKGLGTGQGAVSGPPRKPPGTVAGTARGPEGGSTAQVPAP
TASPPPEGPVLTFQSEKMKGMKELLVATKINSSAIKLQLTAQSQVQMKKQKVSTPSDYTL
SFLKRQRKGL
|
|
|
|---|
| BDBM50582738 |
|---|
| n/a |
|---|
| Name | BDBM50582738 |
|---|
| Synonyms: | CHEMBL5071412 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H51FN4O4S |
|---|
| Mol. Mass. | 666.889 |
|---|
| SMILES | [H][C@@]1(CCC[C@@H]1NC(=O)OC)[C@@](CN1CCCC1)(C1CCN(CC2CN(C2)c2ccc(cc2)S(=O)(=O)C2CC2)CC1)c1cccc(F)c1 |r| |
|---|
| Structure | 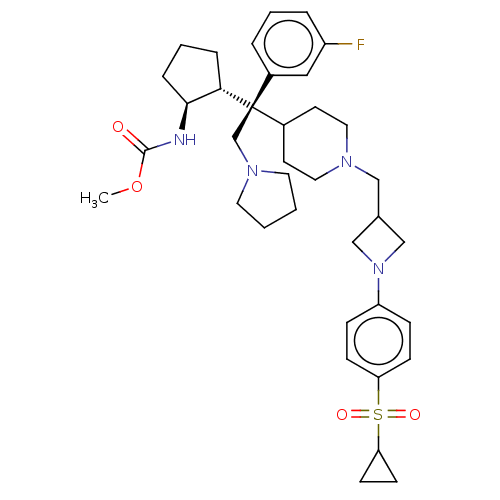
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhang, M; Aguilar, A; Xu, S; Huang, L; Chinnaswamy, K; Sleger, T; Wang, B; Gross, S; Nicolay, BN; Ronseaux, S; Harvey, K; Wang, Y; McEachern, D; Kirchhoff, PD; Liu, Z; Stuckey, J; Tron, AE; Liu, T; Wang, S Discovery of M-1121 as an Orally Active Covalent Inhibitor of Menin-MLL Interaction Capable of Achieving Complete and Long-Lasting Tumor Regression. J Med Chem64:10333-10349 (2021) [PubMed] Article
Zhang, M; Aguilar, A; Xu, S; Huang, L; Chinnaswamy, K; Sleger, T; Wang, B; Gross, S; Nicolay, BN; Ronseaux, S; Harvey, K; Wang, Y; McEachern, D; Kirchhoff, PD; Liu, Z; Stuckey, J; Tron, AE; Liu, T; Wang, S Discovery of M-1121 as an Orally Active Covalent Inhibitor of Menin-MLL Interaction Capable of Achieving Complete and Long-Lasting Tumor Regression. J Med Chem64:10333-10349 (2021) [PubMed] Article