

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Cannabinoid receptor 1 | ||
| Ligand | BDBM50161557 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_306671 (CHEMBL832272) | ||
| IC50 | 190±n/a nM | ||
| Citation |  Plummer, CW; Finke, PE; Mills, SG; Wang, J; Tong, X; Doss, GA; Fong, TM; Lao, JZ; Schaeffer, MT; Chen, J; Shen, CP; Stribling, DS; Shearman, LP; Strack, AM; Van der Ploeg, LH Synthesis and activity of 4,5-diarylimidazoles as human CB1 receptor inverse agonists. Bioorg Med Chem Lett15:1441-6 (2005) [PubMed] Article Plummer, CW; Finke, PE; Mills, SG; Wang, J; Tong, X; Doss, GA; Fong, TM; Lao, JZ; Schaeffer, MT; Chen, J; Shen, CP; Stribling, DS; Shearman, LP; Strack, AM; Van der Ploeg, LH Synthesis and activity of 4,5-diarylimidazoles as human CB1 receptor inverse agonists. Bioorg Med Chem Lett15:1441-6 (2005) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Cannabinoid receptor 1 | |||
| Name: | Cannabinoid receptor 1 | ||
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 52868.96 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P21554 | ||
| Residue: | 472 | ||
| Sequence: |
| ||
| BDBM50161557 | |||
| n/a | |||
| Name | BDBM50161557 | ||
| Synonyms: | 4-(4-Chloro-phenyl)-5-(2,4-dichloro-phenyl)-1-methyl-1H-imidazole-2-carboxylic acid piperidin-1-ylamide | CHEMBL180461 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C22H21Cl3N4O | ||
| Mol. Mass. | 463.787 | ||
| SMILES | Cn1c(nc(c1-c1ccc(Cl)cc1Cl)-c1ccc(Cl)cc1)C(=O)NN1CCCCC1 |(-3.32,1.67,;-2.13,1.09,;-.62,2.16,;.64,1.29,;.21,-.17,;-1.34,-.22,;-2.08,-1.57,;-1.28,-2.87,;-2.02,-4.23,;-3.57,-4.24,;-4.32,-5.6,;-4.36,-2.94,;-3.6,-1.59,;-4.39,-.29,;1.01,-1.51,;2.54,-1.45,;3.33,-2.75,;2.6,-4.12,;3.4,-5.42,;1.05,-4.16,;.26,-2.82,;-.67,3.71,;-2.01,4.46,;.66,4.51,;2.01,3.81,;3.32,3.02,;4.65,3.78,;4.65,5.32,;3.31,6.08,;2.01,5.32,)| | ||
| Structure | 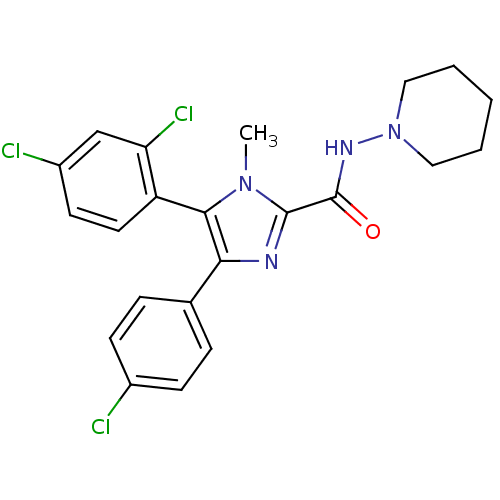
| ||