| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Histone-lysine N-methyltransferase EZH2 |
|---|
| Ligand | BDBM50597877 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2224732 (CHEMBL5138245) |
|---|
| Ki | <0.100000±n/a nM |
|---|
| Citation |  Talukdar, A; Mukherjee, A; Bhattacharya, D Fascinating Transformation of SAM-Competitive Protein Methyltransferase Inhibitors from Nucleoside Analogues to Non-Nucleoside Analogues. J Med Chem65:1662-1684 (2022) [PubMed] Article Talukdar, A; Mukherjee, A; Bhattacharya, D Fascinating Transformation of SAM-Competitive Protein Methyltransferase Inhibitors from Nucleoside Analogues to Non-Nucleoside Analogues. J Med Chem65:1662-1684 (2022) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Histone-lysine N-methyltransferase EZH2 |
|---|
| Name: | Histone-lysine N-methyltransferase EZH2 |
|---|
| Synonyms: | ENX-1 | EZH2 | EZH2_HUMAN | Enhancer of zeste homolog 2 (EZH2) | Histone-lysine N-methyltransferase EZH2 | KMT6 | Lysine N-methyltransferase 6 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 85367.84 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Q15910 |
|---|
| Residue: | 746 |
|---|
| Sequence: | MGQTGKKSEKGPVCWRKRVKSEYMRLRQLKRFRRADEVKSMFSSNRQKILERTEILNQEW
KQRRIQPVHILTSVSSLRGTRECSVTSDLDFPTQVIPLKTLNAVASVPIMYSWSPLQQNF
MVEDETVLHNIPYMGDEVLDQDGTFIEELIKNYDGKVHGDRECGFINDEIFVELVNALGQ
YNDDDDDDDGDDPEEREEKQKDLEDHRDDKESRPPRKFPSDKIFEAISSMFPDKGTAEEL
KEKYKELTEQQLPGALPPECTPNIDGPNAKSVQREQSLHSFHTLFCRRCFKYDCFLHPFH
ATPNTYKRKNTETALDNKPCGPQCYQHLEGAKEFAAALTAERIKTPPKRPGGRRRGRLPN
NSSRPSTPTINVLESKDTDSDREAGTETGGENNDKEEEEKKDETSSSSEANSRCQTPIKM
KPNIEPPENVEWSGAEASMFRVLIGTYYDNFCAIARLIGTKTCRQVYEFRVKESSIIAPA
PAEDVDTPPRKKKRKHRLWAAHCRKIQLKKDGSSNHVYNYQPCDHPRQPCDSSCPCVIAQ
NFCEKFCQCSSECQNRFPGCRCKAQCNTKQCPCYLAVRECDPDLCLTCGAADHWDSKNVS
CKNCSIQRGSKKHLLLAPSDVAGWGIFIKDPVQKNEFISEYCGEIISQDEADRRGKVYDK
YMCSFLFNLNNDFVVDATRKGNKIRFANHSVNPNCYAKVMMVNGDHRIGIFAKRAIQTGE
ELFFDYRYSQADALKYVGIEREMEIP
|
|
|
|---|
| BDBM50597877 |
|---|
| n/a |
|---|
| Name | BDBM50597877 |
|---|
| Synonyms: | CHEMBL5191399 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H24Cl2N2O4 |
|---|
| Mol. Mass. | 451.343 |
|---|
| SMILES | CO[C@H](C1COC1)c1cc(Cl)c2CCN(Cc3c(C)cc(C)[nH]c3=O)C(=O)c2c1Cl |r| |
|---|
| Structure | 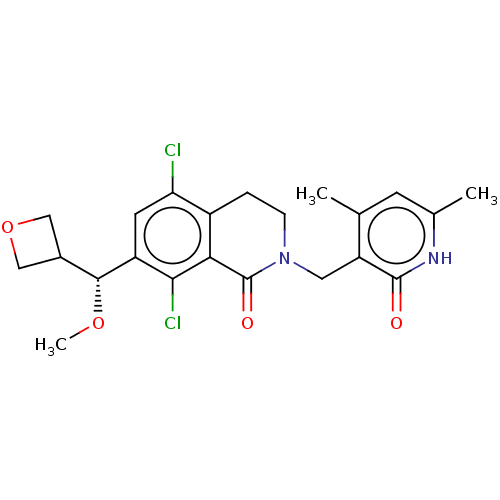
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Talukdar, A; Mukherjee, A; Bhattacharya, D Fascinating Transformation of SAM-Competitive Protein Methyltransferase Inhibitors from Nucleoside Analogues to Non-Nucleoside Analogues. J Med Chem65:1662-1684 (2022) [PubMed] Article
Talukdar, A; Mukherjee, A; Bhattacharya, D Fascinating Transformation of SAM-Competitive Protein Methyltransferase Inhibitors from Nucleoside Analogues to Non-Nucleoside Analogues. J Med Chem65:1662-1684 (2022) [PubMed] Article