| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2D6 |
|---|
| Ligand | BDBM50177530 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_334181 (CHEMBL866475) |
|---|
| IC50 | 2000±n/a nM |
|---|
| Citation |  Berlin, M; Ting, PC; Vaccaro, WD; Aslanian, R; McCormick, KD; Lee, JF; Albanese, MM; Mutahi, MW; Piwinski, JJ; Shih, NY; Duguma, L; Solomon, DM; Zhou, W; Sher, R; Favreau, L; Bryant, M; Korfmacher, WA; Nardo, C; West, RE; Anthes, JC; Williams, SM; Wu, RL; Susan She, H; Rivelli, MA; Corboz, MR; Hey, JA Reduction of CYP450 inhibition in the 4-[(1H-imidazol-4-yl)methyl]piperidine series of histamine H3 receptor antagonists. Bioorg Med Chem Lett16:989-94 (2006) [PubMed] Article Berlin, M; Ting, PC; Vaccaro, WD; Aslanian, R; McCormick, KD; Lee, JF; Albanese, MM; Mutahi, MW; Piwinski, JJ; Shih, NY; Duguma, L; Solomon, DM; Zhou, W; Sher, R; Favreau, L; Bryant, M; Korfmacher, WA; Nardo, C; West, RE; Anthes, JC; Williams, SM; Wu, RL; Susan She, H; Rivelli, MA; Corboz, MR; Hey, JA Reduction of CYP450 inhibition in the 4-[(1H-imidazol-4-yl)methyl]piperidine series of histamine H3 receptor antagonists. Bioorg Med Chem Lett16:989-94 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2D6 |
|---|
| Name: | Cytochrome P450 2D6 |
|---|
| Synonyms: | CP2D6_HUMAN | CYP2D6 | CYP2DL1 | CYPIID6 | Cytochrome P450 2D6 (CYP2D6) | Debrisoquine 4-hydroxylase | P450-DB1 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55774.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P10635 |
|---|
| Residue: | 497 |
|---|
| Sequence: | MGLEALVPLAVIVAIFLLLVDLMHRRQRWAARYPPGPLPLPGLGNLLHVDFQNTPYCFDQ
LRRRFGDVFSLQLAWTPVVVLNGLAAVREALVTHGEDTADRPPVPITQILGFGPRSQGVF
LARYGPAWREQRRFSVSTLRNLGLGKKSLEQWVTEEAACLCAAFANHSGRPFRPNGLLDK
AVSNVIASLTCGRRFEYDDPRFLRLLDLAQEGLKEESGFLREVLNAVPVLLHIPALAGKV
LRFQKAFLTQLDELLTEHRMTWDPAQPPRDLTEAFLAEMEKAKGNPESSFNDENLRIVVA
DLFSAGMVTTSTTLAWGLLLMILHPDVQRRVQQEIDDVIGQVRRPEMGDQAHMPYTTAVI
HEVQRFGDIVPLGVTHMTSRDIEVQGFRIPKGTTLITNLSSVLKDEAVWEKPFRFHPEHF
LDAQGHFVKPEAFLPFSAGRRACLGEPLARMELFLFFTSLLQHFSFSVPTGQPRPSHHGV
FAFLVSPSPYELCAVPR
|
|
|
|---|
| BDBM50177530 |
|---|
| n/a |
|---|
| Name | BDBM50177530 |
|---|
| Synonyms: | 4-((1H-imidazol-4-yl)methyl)-1-(3-(3-(cyclohexyloxy)phenoxy)propyl)piperidine | CHEMBL380139 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H35N3O2 |
|---|
| Mol. Mass. | 397.5536 |
|---|
| SMILES | C(COc1cccc(OC2CCCCC2)c1)CN1CCC(Cc2cnc[nH]2)CC1 |
|---|
| Structure | 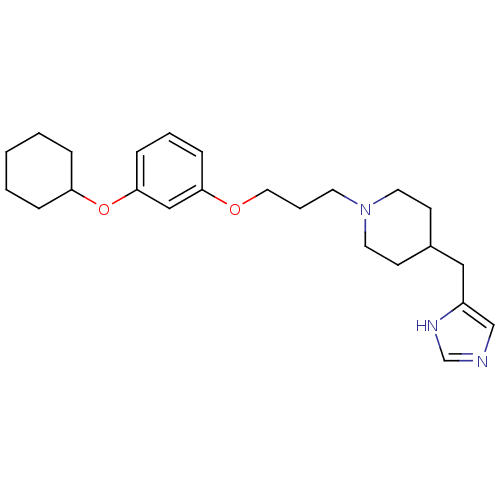
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Berlin, M; Ting, PC; Vaccaro, WD; Aslanian, R; McCormick, KD; Lee, JF; Albanese, MM; Mutahi, MW; Piwinski, JJ; Shih, NY; Duguma, L; Solomon, DM; Zhou, W; Sher, R; Favreau, L; Bryant, M; Korfmacher, WA; Nardo, C; West, RE; Anthes, JC; Williams, SM; Wu, RL; Susan She, H; Rivelli, MA; Corboz, MR; Hey, JA Reduction of CYP450 inhibition in the 4-[(1H-imidazol-4-yl)methyl]piperidine series of histamine H3 receptor antagonists. Bioorg Med Chem Lett16:989-94 (2006) [PubMed] Article
Berlin, M; Ting, PC; Vaccaro, WD; Aslanian, R; McCormick, KD; Lee, JF; Albanese, MM; Mutahi, MW; Piwinski, JJ; Shih, NY; Duguma, L; Solomon, DM; Zhou, W; Sher, R; Favreau, L; Bryant, M; Korfmacher, WA; Nardo, C; West, RE; Anthes, JC; Williams, SM; Wu, RL; Susan She, H; Rivelli, MA; Corboz, MR; Hey, JA Reduction of CYP450 inhibition in the 4-[(1H-imidazol-4-yl)methyl]piperidine series of histamine H3 receptor antagonists. Bioorg Med Chem Lett16:989-94 (2006) [PubMed] Article