| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Kelch-like ECH-associated protein 1 |
|---|
| Ligand | BDBM50601808 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2238230 (CHEMBL5152126) |
|---|
| IC50 | 230±n/a nM |
|---|
| Citation |  Norton, D; Bonnette, WG; Callahan, JF; Carr, MG; Griffiths-Jones, CM; Heightman, TD; Kerns, JK; Nie, H; Rich, SJ; Richardson, C; Rumsey, W; Sanchez, Y; Verdonk, ML; Willems, HMG; Wixted, WE; Wolfe, L; Woolford, AJ; Wu, Z; Davies, TG Fragment-Guided Discovery of Pyrazole Carboxylic Acid Inhibitors of the Kelch-like ECH-Associated Protein 1: Nuclear Factor Erythroid 2 Related Factor 2 (KEAP1:NRF2) Protein-Protein Interaction. J Med Chem64:15949-15972 (2021) [PubMed] Article Norton, D; Bonnette, WG; Callahan, JF; Carr, MG; Griffiths-Jones, CM; Heightman, TD; Kerns, JK; Nie, H; Rich, SJ; Richardson, C; Rumsey, W; Sanchez, Y; Verdonk, ML; Willems, HMG; Wixted, WE; Wolfe, L; Woolford, AJ; Wu, Z; Davies, TG Fragment-Guided Discovery of Pyrazole Carboxylic Acid Inhibitors of the Kelch-like ECH-Associated Protein 1: Nuclear Factor Erythroid 2 Related Factor 2 (KEAP1:NRF2) Protein-Protein Interaction. J Med Chem64:15949-15972 (2021) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Kelch-like ECH-associated protein 1 |
|---|
| Name: | Kelch-like ECH-associated protein 1 |
|---|
| Synonyms: | Cytosolic inhibitor of Nrf2 | INRF2 | KEAP1 | KEAP1/Cullin-3/Microtubule-associated protein tau | KEAP1_HUMAN | KIAA0132 | KLHL19 | Kelch-like protein 19 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 69664.53 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_107924 |
|---|
| Residue: | 624 |
|---|
| Sequence: | MQPDPRPSGAGACCRFLPLQSQCPEGAGDAVMYASTECKAEVTPSQHGNRTFSYTLEDHT
KQAFGIMNELRLSQQLCDVTLQVKYQDAPAAQFMAHKVVLASSSPVFKAMFTNGLREQGM
EVVSIEGIHPKVMERLIEFAYTASISMGEKCVLHVMNGAVMYQIDSVVRACSDFLVQQLD
PSNAIGIANFAEQIGCVELHQRAREYIYMHFGEVAKQEEFFNLSHCQLVTLISRDDLNVR
CESEVFHACINWVKYDCEQRRFYVQALLRAVRCHSLTPNFLQMQLQKCEILQSDSRCKDY
LVKIFEELTLHKPTQVMPCRAPKVGRLIYTAGGYFRQSLSYLEAYNPSDGTWLRLADLQV
PRSGLAGCVVGGLLYAVGGRNNSPDGNTDSSALDCYNPMTNQWSPCAPMSVPRNRIGVGV
IDGHIYAVGGSHGCIHHNSVERYEPERDEWHLVAPMLTRRIGVGVAVLNRLLYAVGGFDG
TNRLNSAECYYPERNEWRMITAMNTIRSGAGVCVLHNCIYAAGGYDGQDQLNSVERYDVE
TETWTFVAPMKHRRSALGITVHQGRIYVLGGYDGHTFLDSVECYDPDTDTWSEVTRMTSG
RSGVGVAVTMEPCRKQIDQQNCTC
|
|
|
|---|
| BDBM50601808 |
|---|
| n/a |
|---|
| Name | BDBM50601808 |
|---|
| Synonyms: | CHEMBL5207900 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C28H37N3O3 |
|---|
| Mol. Mass. | 463.6117 |
|---|
| SMILES | CCCC1CCCCN1C(=O)C1CCCC(C1)c1cccc(c1)-n1ncc(C(O)=O)c1C1CC1 |
|---|
| Structure | 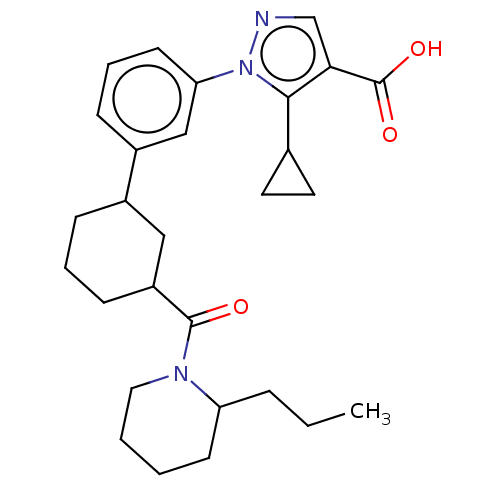
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Norton, D; Bonnette, WG; Callahan, JF; Carr, MG; Griffiths-Jones, CM; Heightman, TD; Kerns, JK; Nie, H; Rich, SJ; Richardson, C; Rumsey, W; Sanchez, Y; Verdonk, ML; Willems, HMG; Wixted, WE; Wolfe, L; Woolford, AJ; Wu, Z; Davies, TG Fragment-Guided Discovery of Pyrazole Carboxylic Acid Inhibitors of the Kelch-like ECH-Associated Protein 1: Nuclear Factor Erythroid 2 Related Factor 2 (KEAP1:NRF2) Protein-Protein Interaction. J Med Chem64:15949-15972 (2021) [PubMed] Article
Norton, D; Bonnette, WG; Callahan, JF; Carr, MG; Griffiths-Jones, CM; Heightman, TD; Kerns, JK; Nie, H; Rich, SJ; Richardson, C; Rumsey, W; Sanchez, Y; Verdonk, ML; Willems, HMG; Wixted, WE; Wolfe, L; Woolford, AJ; Wu, Z; Davies, TG Fragment-Guided Discovery of Pyrazole Carboxylic Acid Inhibitors of the Kelch-like ECH-Associated Protein 1: Nuclear Factor Erythroid 2 Related Factor 2 (KEAP1:NRF2) Protein-Protein Interaction. J Med Chem64:15949-15972 (2021) [PubMed] Article