| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen synthase kinase-3 beta |
|---|
| Ligand | BDBM50186460 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_377353 (CHEMBL865426) |
|---|
| IC50 | 36000±n/a nM |
|---|
| Citation |  Das, J; Furch, JA; Liu, C; Moquin, RV; Lin, J; Spergel, SH; McIntyre, KW; Shuster, DJ; O'Day, KD; Penhallow, B; Hung, CY; Doweyko, AM; Kamath, A; Zhang, H; Marathe, P; Kanner, SB; Lin, TA; Dodd, JH; Barrish, JC; Wityak, J Discovery and SAR of 2-amino-5-(thioaryl)thiazoles as potent and selective Itk inhibitors. Bioorg Med Chem Lett16:3706-12 (2006) [PubMed] Article Das, J; Furch, JA; Liu, C; Moquin, RV; Lin, J; Spergel, SH; McIntyre, KW; Shuster, DJ; O'Day, KD; Penhallow, B; Hung, CY; Doweyko, AM; Kamath, A; Zhang, H; Marathe, P; Kanner, SB; Lin, TA; Dodd, JH; Barrish, JC; Wityak, J Discovery and SAR of 2-amino-5-(thioaryl)thiazoles as potent and selective Itk inhibitors. Bioorg Med Chem Lett16:3706-12 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen synthase kinase-3 beta |
|---|
| Name: | Glycogen synthase kinase-3 beta |
|---|
| Synonyms: | GSK-3 beta | GSK-3, beta | GSK3B | GSK3B_HUMAN | Glycogen synthase kinase 3 beta (GSK3B) | Glycogen synthase kinase 3-beta (GSK3B) | Glycogen synthase kinase-3 beta (GSK-3B) | Glycogen synthase kinase-3 beta (GSK3 Beta) | Glycogen synthase kinase-3 beta (GSK3B) | Glycogen synthase kinase-3B (GSK-3B) | Glycogen synthase kinase-3beta (GSK3B) | Serine/threonine-protein kinase GSK3B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 46756.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49841 |
|---|
| Residue: | 420 |
|---|
| Sequence: | MSGRPRTTSFAESCKPVQQPSAFGSMKVSRDKDGSKVTTVVATPGQGPDRPQEVSYTDTK
VIGNGSFGVVYQAKLCDSGELVAIKKVLQDKRFKNRELQIMRKLDHCNIVRLRYFFYSSG
EKKDEVYLNLVLDYVPETVYRVARHYSRAKQTLPVIYVKLYMYQLFRSLAYIHSFGICHR
DIKPQNLLLDPDTAVLKLCDFGSAKQLVRGEPNVSYICSRYYRAPELIFGATDYTSSIDV
WSAGCVLAELLLGQPIFPGDSGVDQLVEIIKVLGTPTREQIREMNPNYTEFKFPQIKAHP
WTKVFRPRTPPEAIALCSRLLEYTPTARLTPLEACAHSFFDELRDPNVKLPNGRDTPALF
NFTTQELSSNPPLATILIPPHARIQAAASTPTNATAASDANTGDRGQTNNAASASASNST
|
|
|
|---|
| BDBM50186460 |
|---|
| n/a |
|---|
| Name | BDBM50186460 |
|---|
| Synonyms: | CHEMBL209148 | N-(5-(5-(1-acetylpiperazine-4-carbonyl)-4-methoxy-2-methylphenylthio)thiazol-2-yl)-4-((3,3-dimethylbutan-2-ylamino)methyl)benzamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C32H41N5O4S2 |
|---|
| Mol. Mass. | 623.829 |
|---|
| SMILES | COc1cc(C)c(Sc2cnc(NC(=O)c3ccc(CNC(C)C(C)(C)C)cc3)s2)cc1C(=O)N1CCN(CC1)C(C)=O |
|---|
| Structure | 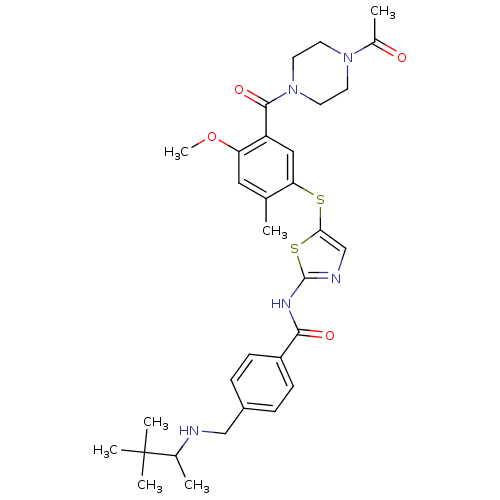
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Das, J; Furch, JA; Liu, C; Moquin, RV; Lin, J; Spergel, SH; McIntyre, KW; Shuster, DJ; O'Day, KD; Penhallow, B; Hung, CY; Doweyko, AM; Kamath, A; Zhang, H; Marathe, P; Kanner, SB; Lin, TA; Dodd, JH; Barrish, JC; Wityak, J Discovery and SAR of 2-amino-5-(thioaryl)thiazoles as potent and selective Itk inhibitors. Bioorg Med Chem Lett16:3706-12 (2006) [PubMed] Article
Das, J; Furch, JA; Liu, C; Moquin, RV; Lin, J; Spergel, SH; McIntyre, KW; Shuster, DJ; O'Day, KD; Penhallow, B; Hung, CY; Doweyko, AM; Kamath, A; Zhang, H; Marathe, P; Kanner, SB; Lin, TA; Dodd, JH; Barrish, JC; Wityak, J Discovery and SAR of 2-amino-5-(thioaryl)thiazoles as potent and selective Itk inhibitors. Bioorg Med Chem Lett16:3706-12 (2006) [PubMed] Article